The latest report from business intelligence provider visiongain offers comprehensive analysis of the global influenza vaccines market. Visiongain assesses that this market will generate revenues of $3.7 billion in 2019.
How this report will benefit you
In this brand new 155-page report you find 64 in-depth tables, charts and graphs – all unavailable elsewhere.
The report provides clear detailed insight into the global Influenza market. Discover the key drivers and challenges affecting the market.
By ordering and reading our brand new report today you stay better informed and ready to act.
Report Scope
• Visiongain’s new investigation shows revenue predictions at overall world market, submarket, company, product and national level.
• This study gives individual analysis for two influenza vaccine submarkets, which are further divided by vaccine technologies:
• Trivalent Influenza Vaccines (TIV)
• Quadrivalent Influenza Vaccines (QIV)
• Our new analysis also shows you revenue predictions for the vaccines produced by these five market leaders: and their market share
• AstraZeneca
• GlaxoSmithKline
• Sanofi Pasteur
• Seqirus
• Others
• Our report analyses the vaccines in research and development and assess their prospects once they enter the market. The R&D pipeline discusses:
• Seasonal Influenza vaccines in development
• Universal Influenza vaccines in development
• The report includes forecasts to 2029 and analysis of pipeline developments for the following specific vaccines:
• Fluzone / VaxiGrip
• Seqirus Influenza Vaccines
• Fluarix / FluLaval
• FluMist / Fluenz
• FluBlok
• Other
• This study shows you forecasts to 2029 of influenza vaccines in developed and developing regions:
• United States
• Japan
• The EU 5 – Germany, France, United Kingdom, Italy and Spain.
• Brazil, Russia, India and China.
• Key Questions Answered by this Report:
• What is the current size of the total global influenza vaccines market? How much will this market be worth from 2019-2029?
• What are the main drivers and restraints that will shape the overall influenza vaccines market over the next ten years?
• What are the main segments within the overall influenza vaccines market? How much will each of these segments be worth for the period 2019-2029? How will the composition of the market change during that time, and why?
• What factors will affect that industry and market over the next ten years?
• What are the largest national markets for influenza vaccines? What is their current status and how will they develop over the next ten years? What are their forecasts for 2019-2029?
• How will political and regulatory factors influence the regional markets?
• How will the market shares of the national markets change by 2029, and which geographical region will lead the market in 2029?
• Who are the leading companies and what are their prospects over the forecast period?
• What are the predictions for existing players and the prospects for new market entrants?
• What are the leading vaccines? What are their revenues and latest developments?
• What are some of the most prominent influenza vaccines currently in development?
• What are the main trends that will affect the influenza vaccines market between 2019 and 2029?
• What are the main strengths, weaknesses, opportunities and threats for the market?
• What are the social, technological, economic and political influences that will shape that industry over the next ten years?
• How will the global influenza vaccines market evolve over the forecasted period, 2019-2029?
Visiongain’s study is intended for anyone requiring commercial analyses for the influenza vaccines market and leading companies. You find data, trends and predictions.
Buy our report today Influenza Vaccines Market Analysis: Forecasts for Trivalent Influenza Vaccines (TIV) & Quadrivalent Influenza Vaccines (QIV), Revenue Forecasts and Analysis of Pipeline Developments for Fluzone / VaxiGrip, Seqirus, Fluarix / FluLaval, FluMist / Fluenz, FluBlok and Other. Avoid missing out by staying informed – get our report now.
Visiongain is a trading partner with the US Federal Government
CCR Ref number: KD4R6
Do you have any custom requirements we can help you with? Any need for specific country, geo region, market segment or specific company information? Contact us today, we can discuss your needs and see how we can help: sara.peerun@visiongain.com
1. Report Overview
1.1 Global Influenza Vaccine Market Overview
1.2 Why You Should Read This Report
1.3 How This Report Delivers
1.4 Main Questions Answered by This Analytical Study
1.5 Who is This Report For?
1.6 Methods of Research and Analysis
1.7 Frequently Asked Questions (FAQ)
1.8 Associated Visiongain Reports
1.9 About Visiongain
2. Introduction to the World Influenza Vaccine Market
2.1 What is Influenza?
2.2 Bird Flu
2.3 The Influenza Virus Kills over 500,000 People Every Year
2.4 2018-2019 Vaccine
2.5 Influenza has a Huge Social and Economic Burden on all Societies
2.6 The Influenza Virus Mutates Annually Rendering the Previous Year’s Vaccines Ineffective
2.6.1 Influenza A
2.6.2 Influenza B
2.6.3 Influenza C
2.6.4 All Current Influenza Vaccines are either Trivalent (TIV) or Quadrivalent (QIV)
2.7 Types of Influenza Vaccines
2.7.1 The Flu Shot
2.7.2 Quadrivalent flu vaccine
2.7.3 Intradermal Influenza Vaccines
2.7.4 High-Dose Influenza Vaccines
2.7.5 Cell based Flu vaccines
2.7.6 Recombinant Influenza Vaccines
2.7.7 Flu Vaccination by jet injectors
2.7.8 Adjuvanted Vaccines
2.7.9 Live attenuated Influenza Vaccine (LAIV)/Nasal Spray Vaccines
3. Influenza Vaccine by technology
3.1 Egg-Based Flu Vaccine
3.2 Cell-Based Flu Vaccines
3.3 Recombinant Flu Vaccines
3.3.1 Fact Sheets of recombinant Influenza Vaccine Flublok
3.4 The Antigenic Drift Mutation Causes Epidemics
3.5 The Antigenic Shift Mutation Causes Pandemics
3.6 An Epidemic or a Pandemic Outbreak?
3.7 Seasonal Influenza Epidemic (Local)
3.8 Pandemic Influenza (Worldwide): ‘A’ Subtype
3.9 Flu Vaccine Effectiveness
3.10 Key Influencers of the Influenza Vaccine Market
3.10.1 Governments and International Organisations
3.10.1.1 Every Year the World Health Organisation Identifies the Most Likely Strain of the Influenza Virus
3.10.1.2 The Governmental Stance of the Importance of Influenza Directly Drives the Market
3.10.2 The WHO’s “Influenza Vaccine Production Transfer Project” Affects Big Pharma’s Vaccine Sales
4. Leading Manufacturers
4.1 Sanofi Pasteur
4.1.1 Market Leader
4.2 Seqirus
4.2.1 CSL Has Bought Novartis's Flu Vaccine Business Forming a New Influenza Vaccine Company Seqirus
4.3 Glaxo SmithKline
4.3.1 GSK is Now Third Placed in the Market
4.4 AstraZeneca
4.4.1 AstraZeneca’s Flumist Product is in Trouble
4.5 Abbott Laboratories
4.6 Sinovac Biotech
5. Influenza Vaccine World Market Outlook, 2019-2029
5.1 The World Influenza Market Performance, 2018
5.1.1 Growing Elderly Population
5.1.2 Paediatric Influenza Vaccines – High Demand Markets
5.1.3 Emerging Markets
5.2 The World Influenza Market Revenue Forecast for 2019-2029
5.3 The World Influenza Market: Industry Trends, 2019-2029
5.3.1 Demand Driven Trends
5.3.2 Supply Efficiency Affects Sales
5.3.3 Funding Trends
5.3.4 Protecting the Workforce of an Economy
5.4 TIV Vaccines will be replaced by QIV Vaccines in the Near Future
5.4.1 Trivalent Influenza Vaccines (TIV)
5.4.2 Quadrivalent Influenza Vaccines (QIV)
5.4.3 Influenza Vaccines by TIV and QIV Type
6. Global Influenza Vaccines Market by Vaccine Technology 2019-2029
6.1 The Influenza Vaccine Market Forecast 2019-2029 by Vaccine Technology (Trivalent and Quadrivalent)
7. Leading Influenza Vaccines 2019-2029
7.1 Sanofi Pasteur Influenza Vaccine Portfolio - Sales Forecast and Discussion 2018-2029
7.2 Seqirus Vaccine Portfolio - Sales Forecast and Discussion 2019-2029
7.3 GSK Influenza Vaccine Portfolio - Sales Forecast and Discussion 2018-2029
7.4 AstraZeneca Influenza Vaccine - Sales Forecast and Discussion 2018-2029
7.5 Sanofi Pasteur has acquired Protein Sciences Company
7.5.1 Flubok Influenza Vaccine - Sales Forecast and Discussion 2019-2029
7.6 Other Vaccines: Sales Forecast and Discussion 2019-2029
8. Influenza Vaccines in Leading Developed Markets, 2019-2029
8.1 Influenza Vaccines: Leading Markets, 2019, 2024, 2029
8.1.1 Influenza Vaccine: Leading Developed Markets
8.2 The US Influenza Vaccine Market: 2019-2029
8.2.1 The US Influenza Vaccine Market: 2018
8.2.2 The US Influenza Vaccine Market Forecast: 2019-2029
8.2.3 Obama Healthcare Reform – the Benefits for Vaccine Sales
8.2.4 In the US Influenza Vaccines Come Equally from Manufacturers and Distributors
8.3 The Japanese Influenza Vaccine Market 2019-2029
8.3.1 The Japanese Influenza Vaccine Market 2018
8.3.2 The Japanese Influenza Vaccine Market Forecast 2019-2029
8.3.3 Filing the Vaccine Gap
8.3.4 Kaketsuken Loses Control of the Japanese Vaccine Market
8.4 The United Kingdom Influenza Vaccine Market 2019-2029
8.4.1 The UK Influenza Vaccine Market, 2018
8.4.2 The UK Influenza Vaccine Market, 2019-2029
8.4.3 Sanofi Pasteur’s QIV Vaccine is Authorized for use in the UK
8.5 The German Influenza Vaccine Market: 2019-2029
8.5.1 The German Influenza Vaccine Market, 2018
8.5.2 The German Influenza Vaccine Market: Forecast 2019-2029
8.6 The French Influenza Vaccine Market 2019-2029
8.6.1 The French Influenza Vaccine Market, 2018
8.6.2 The French Influenza Vaccine Market: Forecast 2019-2029
8.6.3 French Healthcare System – Events, Policies and Effects
8.6.4 The French Population Have the Highest Doubt over Vaccine Safety in Europe
8.7 The Italian Influenza Vaccine Market: 2019-2029
8.7.1 The Italian Influenza Vaccine Market: 2018
8.7.2 The Italian Influenza Vaccine Market Forecast: 2019-2029
8.7.3 Uncertain Foundation – Challenges in Public Healthcare
8.8 The Spanish Influenza Vaccine Market 2019-2029
8.8.1 The Spanish Influenza Vaccine Market: 2018
8.8.2 The Spanish Influenza Vaccine Market Forecast: 2019-2029
8.8.3 District Rules – Economic Challenges
9. Influenza Vaccines in Leading Emerging Markets, 2019-2029
9.1 Influenza Vaccines: Leading Markets, 2019, 2024, 2029
9.1.1 Influenza Vaccine: Leading Emerging National Markets
9.2 The Chinese Influenza Vaccine Market: 2019-2029
9.2.1 The Chinese Influenza Vaccine Market: 2018
9.2.2 The Chinese Influenza Vaccine Market Forecast: 2019-2029
9.2.3 High National Demand for Influenza Vaccines
9.3 The Indian Influenza Vaccine Market: 2019-2029
9.3.1 The Indian Influenza Vaccine Market: 2018
9.3.2 The Indian Influenza Vaccine Market Forecast: 2019-2029
9.3.3 Complications of Influenza Vaccines – How Much Progress Possible?
9.4 The Brazilian Influenza Vaccine Market: 2019-2029
9.4.1 The Brazilian Vaccine Market: 2018
9.4.2 The Brazilian Vaccine Market Forecast: 2019-2029
9.5 The Russian Influenza Vaccine Market: 2019-2029
9.5.1 The Russian Influenza Vaccine Market: 2018
9.5.2 The Russian Influenza Vaccine Market Forecast: 2019-2029
9.5.3 Power of the State
10. Research & Development Pipeline for Flu Vaccines
10.1 Seasonal Influenza Vaccines in Development
10.1.1 Sanofi Pasteur
10.1.1.1 Fluzone QIV High Dose
10.1.1.2 VaxiGrip QIV IM (6-35 months)
10.1.1.3 VaxiGrip QIV IM (3 years +)
10.1.2 Seqirus
10.1.2.1 MF59 QIV and TIV Vaccines
10.1.3 Takeda Pharmaceuticals
10.1.3.1 Takeda has Ceased Development of its TAK850 TIV Influenza Vaccine
10.1.4 Mitsubishi Tanabe Pharma
10.1.4.1 Plant Based Virus-Like- Particle Influenza Vaccine
10.2 Universal Influenza Vaccines in Development
11. Qualitative Analysis of the World Influenza Vaccines Industry and Market, 2019 - 2029
11.1 SWOT Analysis of the World Influenza Vaccines Industry and Market, 2018
11.1.1 Strengths
11.1.1.1 Ageing Population
11.1.1.2 Influenza Prevention is Crucial
11.1.1.3 Sales Growth in Emerging National Markets
11.1.1.4 Rising Awareness of the Disease
11.1.2 Weaknesses
11.1.2.1 Supply Chain Costs
11.1.2.2 Time Pressure
11.1.2.3 Barriers to Market Entry
11.1.2.4 Storage and Handling of Influenza Vaccines
11.1.3 Opportunities
11.1.3.1 Strong Research and Development Pipeline
11.1.3.2 Vaccine Delivery – Innovations and Opportunities for Improvements
11.1.3.3 Universal Flu Vaccine
11.1.3.4 Adult Vaccines – High Need
11.1.4 Threats
11.1.4.1 Productivity Gap
11.2 STEP Analysis of the World Influenza Vaccines Industry and Market, 2018
11.2.1 Social Factors
11.2.1.1 Creating a Healthier Society
11.2.1.2 Public Fears of Side-Effects
11.2.2 Technological Forces
11.2.2.1 Advancements in Vaccine Technology
11.2.3 Economic Factors
11.2.3.1 Cost and Time Restraints
11.2.3.2 Emerging Markets Create Demand
11.2.4 Political Factors
11.2.4.1 Influence of Governments
12. Conclusions
12.1 Prevention of Influenza by Use of Flu Shots Rises in Importance
12.2 Japan as a Rising Consumer in the World Influenza Vaccine Market
12.3 Brazil and India Show Robust Growth
12.4 Emergence of a Universal Flu Vaccine
12.5 The US to Retain its Dominance
12.6 Concluding Remarks
Appendices
Associated Reports
Visiongain Report Sales Order Form
About Visiongain
Visiongain Report Evaluation Form
List of Tables
Table 2.1 Seasonal Influenza Vaccine-Total Doses Distributed in the US Market: 2018-2029
Table 2.2 Influenza Vaccine Products for the 2018-2019 Influenza Season
Table 3.1 Influenza Vaccine Effectiveness (%) 2004/5-2015/6
Table 5.1 World Influenza Vaccine Market: Revenue Forecast ($m), Annual Growth (%), CAGR (%), 2018-2029
Table 5.2 The World Market for Influenza Vaccines by Pharmaceutical Companies and ‘Others’; Revenue Forecast ($m), AGR (%) Market Share (%), 2018- 2029
Table 5.3 Influenza Vaccines Divided into TIV and QIV Type, 2018
Table 6.1 The Influenza Vaccine Revenue Forecast ($m), Annual Growth (%), CAGR (%) by Vaccine Technology, 2019-2029
Table 7.1 Leading Influenza Vaccines: Revenues ($m), 2018
Table 7.2 Fluzone/VaxiGrip: Historical Revenue ($m), 2013-2015
Table 7.3 Sanofi Pasteur (Fluzone/VaxiGrip); Revenue Forecast ($m), Annual Growth (%), CAGR (%), 2018-2029
Table 7.4 Seqirus Influenza Vaccines; Revenue Forecast ($m), Annual Growth (%), CAGR (%), 2019-2029
Table 7.5 Fluarix/FluLaval: Historical Revenue ($m), 2013-2015
Table 7.6 Fluarix/FluLaval: Revenue Forecast ($m), Annual Growth ($m), CAGR (%), 2019-2029
Table 7.7 FluMist/Fluenz: Revenue ($m), 2013-2015
Table 7.8 FluMist/Fluenz: Revenue Forecast ($m), Annual Growth (%), CAGR (%), 2019-2029
Table 7.9 FluBlok Historical Revenue ($m), 2013-2014
Table 7.10 FluBlok: Revenue Forecast ($m), Annual Growth (%), CAGR (%), 2019-2029
Table 7.11 Other Influenza Vaccines Forecast: ($m), Annual Growth (%), CAGR (%), 2019-2029
Table 8.1 Influenza Vaccines Market Revenues ($m) and Market Shares (%) by Leading Country, 2019, 2024 and 2029
Table 8.2 The US Influenza Vaccine Revenue Forecast ($m), Annual Growth (%), CAGR (%), 2019-2029
Table 8.3 The Japanese Influenza Vaccine Revenue Forecast ($m), Annual Growth (%), CAGR (%), 2019-2029
Table 8.4 The UK Influenza Vaccines Forecast ($m), Annual Growth (%) and CAGR (%), 2019-2029
Table 8.5 The German Influenza Vaccines Forecast ($m), Annual Growth (%) and CAGR (%), 2019-2029
Table 8.6 The French Vaccines Forecast ($m), Annual Growth (%) and CAGR (%), 2019-2029
Table 8.7 The Italian Influenza Vaccines Forecast ($m), Annual Growth (%) and CAGR (%), 2019-2029
Table 8.8 The Spanish Influenza Vaccines Forecast ($m), Annual Growth (%) and CAGR (%), 2019-2029
Table 9.1 Influenza Vaccines Market Revenues ($m) and Market Shares (%) by Leading Country, 2019, 2024 and 2029
Table 9.2 The Chinese Influenza Vaccines Forecast ($bn), Annual Growth (%) and CAGR (%), 2019-2029
Table 9.3 The Indian Influenza Vaccines Forecast ($m), Annual Growth (%) and CAGR (%), 2019-2029
Table 9.4 The Brazilian Influenza Vaccines Forecast ($m), Annual Growth (%) and CAGR (%), 2019-2029
Table 9.5 The Russian Influenza Vaccines Forecast ($m), Annual Growth (%) and CAGR (%), 2018-2028
Table 10.1 Seasonal Influenza Vaccines in Development, 2019
Table 10.2 Universal Influenza Vaccines in Development, 2018
List of Figures
Figure 1.1 Influenza Vaccines Market Segmentation
Figure 5.1 Influenza Vaccine’s Market Share of Global Vaccines Industry (%), 2018
Figure 5.2 Growth in the Uptake of Influenza Vaccines, 1990-2015
Figure 5.3 Influenza Vaccines: Segmentation by Age (%), 2018
Figure 5.4 Influenza Vaccinations by Age, 2018
Figure 5.5 Influenza Vaccines: Market Share by Geographical Region, 2018
Figure 5.6 The World Market for Influenza Vaccines Divided into Pharma Companies and ‘Others’, 2018- 2028
Figure 5.7 Influenza Market Share (%) by Pharma Companies and ‘Others’: 2018 and 2029
Figure 5.8 World Influenza Vaccines Market: Drivers and Restraints, 2019-2029
Figure 6.1 The Global Influenza Vaccine Revenue Forecast ($m) by Vaccine Technology, 2019-2029
Figure 7.1. Afluria Influenza Vaccines; Revenue Forecast ($m), 2018-2029
Figure 7.2. GSK Influenza Vaccines; Revenue Forecast ($m), 2019-2029
Figure 7.3 FluMist/ Fluenz: Revenue Forecast ($m), 2019-2029
Figure 7.4 FluBlok: Revenue Forecast ($m), 2019-2029
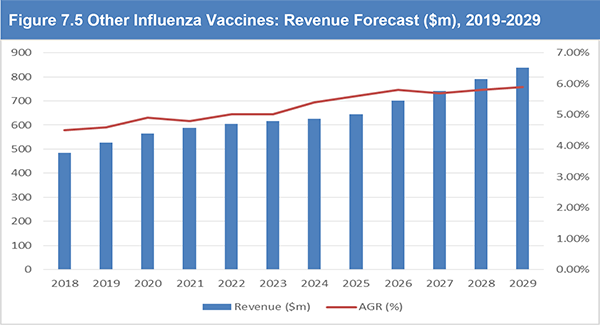
Figure 7.5 Other Influenza Vaccines: Revenue Forecast ($m), 2019-2029
Figure 8.1 Influenza Vaccine Market Share (%) by Country, 2019
Figure 8.2 Developed National Markets: Drivers and Restraints, 2019-2029
Figure 8.3 The US Influenza Vaccine Revenue Forecast ($m), 2019-2029
Figure 8.4 The Japanese Influenza Vaccine Revenue Forecast ($m), 2018-2029
Figure 8.5 The UK Influenza Vaccines Revenue Forecast ($m), 2019-2029
Figure 8.6 The German Influenza Vaccines Revenue Forecast ($m), 2019-2029
Figure 8.7 The French Influenza Vaccines Revenue Forecast ($m), 2019-2029
Figure 8.8 The Italian Influenza Vaccines Revenue Forecast ($m), 2019-2029
Figure 8.9 The Spanish Influenza Vaccines Revenue Forecast ($m), 2019-2029
Figure 9.1 Emerging National Markets: Drivers and Restraints, 2019-2029
Figure 9.2 The Chinese Influenza Vaccines Revenue Forecast ($m), 2019-2029
Figure 9.3 The Indian Influenza Vaccines Revenue Forecast ($m), 2019-2029
Figure 9.4 The Brazilian Influenza Vaccines Revenue Forecast ($m), 2018-2028
Figure 9.5 The Russian Influenza Vaccines Revenue Forecast ($m), 2019-2029
Figure 11.1 SWOT Analysis of the Influenza Vaccines Market, 2018
Figure 11.2 STEP Analysis of the Influenza Vaccines Market, 2018
Advisory Committee for Immunization Practices
Astellas Pharmaceuticals
AstraZeneca
Bharat Immunologicals and Biologicals Corporation Limited
Binnopharm
Bio Farma
bioCSL
BioManguinhos/Oswaldo Cruz Foundation
BiondVax
BiondVax
Bionor
Biremx
Brazilian Ministry of Health
Butantan Institute
Cabtacuzino Institute
Center for Biologics Evaluation and Research
Centers for Disease Control and Prevention (CDC)
Centre for Disease Control and Prevention
Chemo-Sero-Therapeutic Research Institute
China National Biotech Group
China’s National Regulatory Authority
Chinese Food and Drug Administration
ClearPath
CSL
CureVac
Daiichi Sankyo
Developing Countries Vaccine Manufacturers Network (DCVMN)
Dynavax
Flanders Institute
Food and Drug Administration (FDA)
FORT
Fresenius Pharmaceuticals
Gamma Vaccines
Gavi Alliance
Glaxo Smith Klein
GlaxoWellcome
Global Influenza Programme
Governmental Pharmaceutical Organization (GPO)
Green Cross Corporation
Grippol
Health Service Bureau, Japan
Hualan Bio
Immune Targeting Systems
Indian Association of Paediatrics
International Vaccine Access Center (IVAC)
Janssen Pharmaceuticals
Jenner Institute, University of oxford
Johnson & Johnson
Krka Pharmaceuticals
Medicago
Medicare
Medicines and Healthcare products Regulatory Agency (MHRA)
MedImmune
Merck
Mitsubishi Tanabe Pharma
MSD Pharmaceuticals
National Health Service, UK
Netherlands Vaccine Institute
Novartis
Organisation for Economic Co-operation and Development
Panacea Biotech
Partnership for Influenza Vaccine Introduction
Petrovax
Pfizer
Protein Science Corporation
Razi Institute
Sanofi Pasteur
Seqirus
Serum Institute of India (SLL)
Servizio Sanitario Nationale
Shionogi Pharmaceuticals
Sinopharm
Sistema Unico de Saude
SmithKline Beecham
Takeda Pharmaceuticals
Terumo Pharmaceuticals
The Ministry of Health, Labour and Welfare, Japan
The Scripps Research Institute
Torlak Institut
Ultriks
US Department of Health and Human Services
Vacsera
VaxInnate
VaxInnate Corporation
WHO Strategic Advisory Group of Experts
World Health Organization




























