The Contract Manufacturing Outsourcing (CMO) of Sterile Injectable Drugs Market Report 2022-2032: This report will prove invaluable to leading firms striving for new revenue pockets if they wish to better understand the industry and its underlying dynamics. It will be useful for companies that would like to expand into different industries or to expand their existing operations in a new region.
Rising Demand for Outsourcing Drug Manufacturing by Pharmaceutical Companies to Boost Market Growth
Over the last few decades, contract manufacturing organisations (CMOs) have evolved as a viable alternative to pharmaceutical businesses’ in-house research and production departments. Contract manufacturing organisations (CMOs) are proven to be a successful business model as they become more integrated into pharmaceutical firms’ value chains. Despite the fact that the sector as a whole is expanding, many CMOs are encountering new obstacles. As a result of growing expenses, constantly improving technology, and a surge in mergers and acquisitions, competition in this sector has intensified. As a result of these considerations, organisations must decide what actions they might take to defend or improve their position.
Manufacturing costs are much lower in the Asia-Pacific area than in North America and Europe, and favourable laws are fuelling the rise of the CMO business. Despite the fact that API manufacturing services have mostly shifted to China and India, the United States remains the major site for pharmaceutical development outsourcing. Several reasons contribute to this predicament, including the abundance of funding and the concentration of pharmaceutical research hubs in universities. Pharmaceutical development in poor countries is also unfavourable owing to quality difficulties, logistical constraints, and legal challenges.
Market Growth is Hindered by Growing Competition from Small Manufacturers
The market’s discontinuance is considered in the perspective of the whole evaluation process for product lifespan and market viability. Drug discontinuation is defined as authorised products that are removed from the market or withdrawn for reasons other than safety or efficacy and are never again discontinued, marketed, exported, or used in the military. The possibility for a pharmaceutical corporation to pull a medicine from the market can have serious effects, including financial losses, loss of goodwill, and disrupted therapeutic research.
The consumer product literature contains well-developed dominating pricing methods, particularly skimming and penetration pricing. Pharmaceutical items use the same market-dynamic pricing mechanisms as other products. According to the current market study, the primary factor determining a product’s pricing is a larger therapeutic benefit. The greater the clinical benefit over previously accessible medications on the market, the higher the initial price of the product. If the sterile injectables medication product’s life cycle was not extended, this incident might result in significant losses for the corporation and, finally, product termination.
What Questions Should You Ask before Buying a Market Research Report?
• How is the CMO of Sterile Injectable Drugs market evolving?
• What is driving and restraining the CMO of Sterile Injectable Drugs market?
• How will each CMO of Sterile Injectable Drugs submarket segment grow over the forecast period and how much revenue will these submarkets account for in 2032?
• How will the market shares for each CMO of Sterile Injectable Drugs submarket develop from 2022 to 2032?
• What will be the main driver for the overall market from 2022 to 2032?
• Will leading CMO of Sterile Injectable Drugs markets broadly follow the macroeconomic dynamics, or will individual national markets outperform others?
• How will the market shares of the national markets change by 2032 and which geographical region will lead the market in 2032?
• Who are the leading players and what are their prospects over the forecast period?
• What are the CMO of Sterile Injectable Drugs projects for these leading companies?
• How will the industry evolve during the period between 2022 and 2032? What are the implications of CMO of Sterile Injectable Drugs projects taking place now and over the next 10 years?
• Is there a greater need for product commercialisation to further scale the CMO of Sterile Injectable Drugs market?
• Where is the CMO of Sterile Injectable Drugs market heading and how can you ensure you are at the forefront of the market?
• What are the best investment options for new product and service lines?
• What are the key prospects for moving companies into a new growth path and C-suite?
You need to discover how this will impact the CMO of Sterile Injectable Drugs market today, and over the next 10 years:
• Our 357-page report provides 116 tables and 175 charts/graphs exclusively to you.
• The report highlights key lucrative areas in the industry so you can target them – NOW.
• It contains in-depth analysis of global, regional and national sales and growth.
• It highlights for you the key successful trends, changes and revenue projections made by your competitors.
This report tells you TODAY how the CMO of Sterile Injectable Drugs market will develop in the next 10 years, and in line with the variations in COVID-19 economic recession and bounce. This market is more critical now than at any point over the last 10 years.
Forecasts to 2032 and other analyses reveal commercial prospects
• In addition to revenue forecasting to 2032, our new study provides you with recent results, growth rates, and market shares.
• You will find original analyses, with business outlooks and developments.
• Discover qualitative analyses (including market dynamics, drivers, opportunities, restraints and challenges), cost structure, impact of rising CMO of Sterile Injectable Drugs prices and recent developments.
This report includes data analysis and invaluable insight into how COVID-19 will affect the industry and your company. Four COVID-19 recovery patterns and their impact, namely, “V”, “L”, “W” and “U” are discussed in this report.
Segments Covered in the Report
Molecule Type
• Small Molecule
• Large Molecule
Type
• Monoclonal Antibodies (mAbs)
• Cytokines
• Insulin
• Peptide Hormones
• Vaccines
• Immunoglobulins
• Blood Factors
• Peptide Antibiotics
• Others
Application
• Cancer
• Diabetes
• Cardiovascular diseases
• CNS
• Infectious
• Others
Container Type
• Bottles
• Ampoules
• Vials
• Prefilled syringes
• Bags
Route of Administration
• Subcutaneous (SC)
• Intravenous (IV)
• Intramuscular (IM)
• Others

In addition to the revenue predictions for the overall world market and segments, you will also find revenue forecasts for five regional and 18 leading national markets:
North America
• U.S.
• Canada
Europe
• Germany
• UK
• France
• Spain
• Italy
• Russia
• Rest of Europe
Asia Pacific
• China
• Japan
• India
• Australia
• South Korea
• Rest of Asia Pacific
Latin America
• Brazil
• Mexico
• Rest of Latin America
MEA
• GCC
• South Africa
• Rest of MEA
Need industry data? Please contact us today.
The report also includes profiles and for some of the leading companies in the Contract Manufacturing Outsourcing (CMO) of Sterile Injectable Drugs Market, 2022 to 2032, with a focus on this segment of these companies’ operations.
Leading companies and the potential for market growth
• Adare Pharma Solutions
• Aenova Group
• Almac Group
• Avara Pharmaceutical Services, Inc.
• Baxter International Inc.
• Boehringer Ingelheim International GmbH
• Catalent, Inc.
• CordenPharma
• Eli Lilly and Company
• Evonik Industries AG
• F. Hoffmann-La Roche Ltd
• FAMAR Health Care Services
• Fresenius Kabi Ag
• Grifols SA
• Hikma Pharmaceuticals PLC
• Jubilant Pharmova Limited
• Lonza
• Nexus Pharmaceuticals, Inc.
• Patheon, Inc. (Thermo Fischer)
• Pfizer CentreOne
• Recipharm AB
• Siegfried Holding AG
Overall world revenue for Contract Manufacturing Outsourcing (CMO) of Sterile Injectable Drugs Market, 2022 to 2032 in terms of value the market will surpass US$22,300.0 million in 2023, our work calculates. We predict strong revenue growth through to 2032. Our work identifies which organizations hold the greatest potential. Discover their capabilities, progress, and commercial prospects, helping you stay ahead.
How will the Contract Manufacturing Outsourcing (CMO) of Sterile Injectable Drugs Market, 2022 to 2032 report help you?
In summary, our 370+ page report provides you with the following knowledge:
• Revenue forecasts to 2032 for Contract Manufacturing Outsourcing (CMO) of Sterile Injectable Drugs Market, 2022 to 2032 Market, with forecasts for molecule type, type, application, container type, and route of administration, each forecast at a global and regional level – discover the industry’s prospects, finding the most lucrative places for investments and revenues.
• Revenue forecasts to 2032 for five regional and 18 key national markets – See forecasts for the Contract Manufacturing Outsourcing (CMO) of Sterile Injectable Drugs Market, 2022 to 2032 market in North America, Europe, Asia-Pacific, LA, and MEA. Also forecasted is the market in the US, Canada, Mexico, Brazil, Germany, France, UK, Italy, China, India, Japan, and Australia among other prominent economies.
• Prospects for established firms and those seeking to enter the market – including company profiles for 22 of the major companies involved in the Contract Manufacturing Outsourcing (CMO) of Sterile Injectable Drugs Market, 2022 to 2032.
Find quantitative and qualitative analyses with independent predictions. Receive information that only our report contains, staying informed with invaluable business intelligence.
Information found nowhere else
With our new report, you are less likely to fall behind in knowledge or miss out on opportunities. See how our work could benefit your research, analyses, and decisions. Visiongain’s study is for everybody needing commercial analyses for the Contract Manufacturing Outsourcing (CMO) of Sterile Injectable Drugs Market, 2022 to 2032, market-leading companies. You will find data, trends and predictions.
To access the data contained in this document please email contactus@visiongain.com
Buy our report today Contract Manufacturing Outsourcing (CMO) of Sterile Injectable Drugs Market Report 2022-2032: Forecasts by Molecule Type (Small Molecule, Large Molecule), by Type (Monoclonal Antibodies (mAbs), Cytokines, Insulin, Peptide Hormones, Vaccines, Immunoglobulins, Blood Factors, Peptide Antibiotics, Others), by Application (Cancer, Diabetes, Cardiovascular Diseases, CNS, Infectious, Others), by Container Type (Bottles, Ampoules, Vials, Pre-filled Syringes, Bags), by Route of Administration (Subcutaneous (SC), Intravenous (IV), Intramuscular (IM), Others) AND Regional and Leading National Market Analysis PLUS Analysis of Leading Companies AND COVID-19 Impact and Recovery Pattern Analysis. Avoid missing out by staying informed – order our report now.
Visiongain is a trading partner with the US Federal Government
CCR Ref number: KD4R6
Do you have any custom requirements we can help you with? Any need for a specific country, geo region, market segment or specific company information? Contact us today, we can discuss your needs and see how we can help: contactus@visiongain.com
1. Report Overview
1.1 Objectives of the Study
1.2 Introduction to CMO of Sterile Injectable Drugs Market
1.3 What This Report Delivers
1.4 Why You Should Read This Report
1.5 Key Questions Answered by This Analytical Report Include:
1.6 Who is This Report For?
1.7 Methodology
1.7.1 Market Definitions
1.7.2 Market Evaluation & Forecasting Methodology
1.7.3 Data Validation
1.7.3.1 Primary Research
1.7.3.2 Secondary Research
1.8 Frequently Asked Questions (FAQs)
1.9 Associated Visiongain Reports
1.10 About Visiongain
2 Executive Summary
3 Market Overview
3.1 Key Findings
3.2 Market Dynamics
3.2.1 Market Driving Factors
3.2.1.1 Growing Focus on Development to Treat Cancer
3.2.1.2 Rising Prevalence of Chronic Diseases
3.2.1.3 Rapid FDA Approvals of Sterile Injectable Drugs to Fuel Market Growth
3.2.1.4 As Manufacturers Invest in New Manufacturing Facilities, The Demand for Sterile Injectables is Expected to Rise
3.2.1.5 Increasing Demand for Biologicals is Expected to Drive the Demand
3.2.2 Market Restraining Factors
3.2.2.1 Growing Competition from Small Manufacturers
3.2.2.2 High Operational Costs
3.2.2.3 Inadequate Healthcare Infrastructure Across Developing Economies Challenging Market Growth
3.2.3 Market Opportunities
3.2.3.1 Prefilled Syringes to Offer Lucrative Growth Prospects
3.2.3.2 Growing Investments
3.2.3.3 Expanding Clinical Trial Opportunities in the Asia Pacific Region
3.2.4 Market Challenges
3.2.4.1 Disruption of Clinical Trials Due to Pandemic Breakout
3.3 COVID-19 Impact Analysis
3.4 Porter’s Five Forces Analysis
3.4.1 Supplier Power
3.4.2 Buyer Power
3.4.3 Competitive Rivalry
3.4.4 Threat from Substitutes
3.4.5 Threat of New Entrants
3.5 PEST Analysis
3.5.1 Political Factors Impacting CMO of Sterile Injectable Drugs Market
3.5.2 Economic Factors Impacting CMO of Sterile Injectable Drugs Market
3.5.3 Social Factors Impacting CMO of Sterile Injectable Drugs Market
3.5.4 Technological Factors Impacting CMO of Sterile Injectable Drugs Market
4 CMO of Sterile Injectable Drugs Market Analysis by Molecule Type
4.1 Key Findings
4.2 Molecule Type Segment: Market Attractiveness Index
4.3 CMO of Sterile Injectable Drugs Market Share by Molecule Type, 2022 & 2032
4.4 Small Molecule
4.5 Large Molecule
4.6 CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Molecule Type
5 CMO of Sterile Injectable Drugs Market Analysis by Type
5.1 Key Findings
5.2 Type Segment: Market Attractiveness Index
5.3 CMO of Sterile Injectable Drugs Market Share by Type, 2022 & 2032
5.4 Monoclonal Antibodies (mAbs)
5.5 Cytokines
5.6 Insulin
5.7 Peptide Hormones
5.8 Vaccines
5.9 Immunoglobulins
5.10 Blood Factor
5.11 Peptide Antibiotics
5.12 Others
5.13 CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Type
6 CMO of Sterile Injectable Drugs Market Analysis by Application
6.1 Key Findings
6.2 Application Segment: Market Attractiveness Index
6.3 CMO of Sterile Injectable Drugs Market Share by Application, 2022 & 2032
6.4 Cancer
6.5 Diabetes
6.6 Cardiovascular Diseases
6.7 CNS
6.8 Infectious
6.9 Others
6.10 CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Application
7 CMO of Sterile Injectable Drugs Market Analysis by Container Type
7.1 Key Findings
7.2 Container Type Segment: Market Attractiveness Index
7.3 CMO of Sterile Injectable Drugs Market Share by Container Type, 2022 & 2032
7.4 Bottles
7.5 Ampoules
7.6 Vials
7.7 Prefilled Syringes
7.8 Bags
7.9 CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Container Type
8 CMO of Sterile Injectable Drugs Market Analysis by Route of Administration
8.1 Key Findings
8.2 Route of Administration Segment: Market Attractiveness Index
8.3 CMO of Sterile Injectable Drugs Market Share by Route of Administration, 2022 & 2032
8.4 Subcutaneous
8.5 Intravenous
8.6 Intramuscular
8.7 Others
8.8 CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Route of Administration
9 CMO of Sterile Injectable Drugs Market Analysis by Service
9.1 Key Findings
9.2 Service Segment: Market Attractiveness Index
9.3 CMO of Sterile Injectable Drugs Market Share by Service, 2022 & 2032
9.4 Bioanalytical Testing
9.5 Method Development & Validation
9.6 Stability Testing
9.7 Others
9.8 CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Service
10 CMO of Sterile Injectable Drugs Market Analysis by Region
10.1 Key Findings
10.2 Regional Market Size Estimation and Forecast
11 North America CMO of Sterile Injectable Drugs Market Analysis
11.1 Key Findings
11.2 North America CMO of Sterile Injectable Drugs Market Attractiveness Index
11.3 North America CMO of Sterile Injectable Drugs Market by Country, 2022, 2027 & 2032 (US$ mn)
11.4 North America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Country
11.5 North America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Molecule Type
11.6 North America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Type
11.7 North America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Application
11.8 North America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Container Type
11.9 North America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Route of Administration
11.10 North America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Service
11.11 U.S.
11.12 Canada
12 Europe CMO of Sterile Injectable Drugs Market Analysis
12.1 Key Findings
12.2 Europe CMO of Sterile Injectable Drugs Market Attractiveness Index
12.3 Europe CMO of Sterile Injectable Drugs Market by Country, 2022, 2027 & 2032 (US$ mn)
12.4 Europe CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Country
12.5 Europe CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Molecule Type
12.6 Europe CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Type
12.7 Europe CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Application
12.8 Europe CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Container Type
12.9 Europe CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Route of Administration
12.10 Europe CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Service
12.11 Germany
12.12 U.K.
12.13 France
12.14 Italy
12.15 Spain
12.16 Russia
12.17 Rest of Europe
13 Asia Pacific CMO of Sterile Injectable Drugs Market Analysis
13.1 Key Findings
13.2 Asia Pacific CMO of Sterile Injectable Drugs Market Attractiveness Index
13.3 Asia Pacific CMO of Sterile Injectable Drugs Market by Country, 2022, 2027 & 2032 (US$ mn)
13.4 Asia Pacific CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Country
13.5 Asia Pacific CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Molecule Type
13.6 Asia Pacific CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Type
13.7 Asia Pacific CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Application
13.8 Asia Pacific CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Container Type
13.9 Asia Pacific CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Route of Administration
13.10 Asia Pacific CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Service
13.11 China
13.12 Japan
13.13 India
13.14 Australia
13.15 South-East Asia
13.16 Rest of Asia Pacific
14 Latin America CMO of Sterile Injectable Drugs Market Analysis
14.1 Key Findings
14.2 Latin America CMO of Sterile Injectable Drugs Market Attractiveness Index
14.3 Latin America CMO of Sterile Injectable Drugs Market by Country, 2022, 2027 & 2032 (US$ mn)
14.4 Latin America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Country
14.5 Latin America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Molecule Type
14.6 Latin America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Type
14.7 Latin America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Application
14.8 Latin America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Container Type
14.9 Latin America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Route of Administration
14.10 Latin America CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Service
14.11 Brazil
14.12 Mexico
14.13 Rest of Latin America
15 Middle East & Africa CMO of Sterile Injectable Drugs Market Analysis
15.1 Key Findings
15.2 Middle East & Africa CMO of Sterile Injectable Drugs Market Attractiveness Index
15.3 Middle East & Africa CMO of Sterile Injectable Drugs Market by Country, 2022, 2027 & 2032 (US$ mn)
15.4 Middle East & Africa CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Country
15.5 Middle East & Africa CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Molecule Type
15.6 Middle East & Africa CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Type
15.7 Middle East & Africa CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Application
15.8 Middle East & Africa CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Container Type
15.9 Middle East & Africa CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Route of Administration
15.10 Middle East & Africa CMO of Sterile Injectable Drugs Market Size Estimation and Forecast by Service
15.11 GCC
15.12 South Africa
15.13 Rest of Middle East & Africa
16 Competitive Landscape
16.1 Company Share Analysis
16.2 Key Business Strategy Analysis
17 Company Profiles
17.1 Aenova Group
17.1.1 Company Snapshot
17.1.2 Company Overview
17.1.3 Product Benchmarking
17.1.4 Strategic Outlook
17.2 Almac Group
17.2.1 Company Snapshot
17.2.2 Company Overview
17.2.3 Product Benchmarking
17.2.4 Strategic Outlook
17.3 Baxter International Inc.
17.3.1 Company Snapshot
17.3.2 Company Overview
17.3.3 Financial Analysis
17.3.3.1 Net Revenue, 2017-2021
17.3.3.2 Regional Revenue/Market Shares, 2020
17.3.4 Product Benchmarking
17.3.5 Strategic Outlook
17.4 Boehringer Ingelheim International GmbH
17.4.1 Company Snapshot
17.4.2 Company Overview
17.4.3 Financial Analysis
17.4.3.1 Net Revenue, 2017-2021
17.4.3.2 Regional Revenue/Market Shares, 2021
17.4.3.3 Segmental Revenue/Market Shares, 2021
17.4.4 Product Benchmarking
17.5 Catalent, Inc.
17.5.1 Company Snapshot
17.5.2 Company Overview
17.5.3 Financial Analysis
17.5.3.1 Net Revenue, 2017-2021
17.5.3.2 Regional Revenue/Market Shares, 2021
17.5.3.3 Segmental Revenue/Market Shares, 2021
17.5.4 Product Benchmarking
17.5.5 Strategic Outlook
17.6 CordenPharma
17.6.1 Company Snapshot
17.6.2 Company Overview
17.6.3 Product Benchmarking
17.6.4 Strategic Outlook
17.7 Evonik Industries AG
17.7.1 Company Snapshot
17.7.2 Company Overview
17.7.3 Financial Analysis
17.7.3.1 Net Revenue, 2017-2021
17.7.3.2 Regional Revenue/Market Shares, 2021
17.7.3.3 Segmental Revenue/Market Shares, 2021
17.7.4 Product Benchmarking
17.7.5 Strategic Outlook
17.8 FAMAR Health Care Services
17.8.1 Company Snapshot
17.8.2 Company Overview
17.8.3 Product Benchmarking
17.8.4 Strategic Outlook
17.9 Fresenius Kabi Ag
17.9.1 Company Snapshot
17.9.2 Company Overview
17.9.3 Financial Analysis
17.9.3.1 Net Revenue, 2017-2021
17.9.3.2 Regional Revenue/Market Shares, 2021
17.9.3.3 Segmental Revenue/Market Shares, 2021
17.9.4 Product Benchmarking
17.9.5 Strategic Outlook
17.10 Grifols SA
17.10.1 Company Snapshot
17.10.2 Company Overview
17.10.3 Financial Analysis
17.10.3.1 Net Revenue, 2017-2021
17.10.3.2 Regional Revenue/Market Shares, 2021
17.10.3.3 Segmental Revenue/Market Shares, 2021
17.10.4 Product Benchmarking
17.10.5 Strategic Outlook
17.11 Hikma Pharmaceuticals PLC
17.11.1 Company Snapshot
17.11.2 Company Overview
17.11.3 Financial Analysis
17.11.3.1 Net Revenue, 2017-2021
17.11.3.2 Regional Revenue/Market Shares, 2021
17.11.3.3 Segmental Revenue/Market Shares, 2021
17.11.4 Product Benchmarking
17.11.5 Strategic Outlook
17.12 Jubilant Pharmova Limited
17.12.1 Company Snapshot
17.12.2 Company Overview
17.12.3 Financial Analysis
17.12.3.1 Net Revenue, 2017-2021
17.12.3.2 Regional Revenue/Market Shares, 2021
17.12.3.3 Segmental Revenue/Market Shares, 2021
17.12.4 Product Benchmarking
17.12.5 Strategic Outlook
17.13 Lonza
17.13.1 Company Snapshot
17.13.2 Company Overview
17.13.3 Financial Analysis
17.13.3.1 Net Revenue, 2017-2021
17.13.3.2 Regional Revenue/Market Shares, 2021
17.13.3.3 Segmental Revenue/Market Shares, 2021
17.13.4 Product Benchmarking
17.13.5 Strategic Outlook
17.14 Patheon, Inc. (Thermo Fischer)
17.14.1 Company Snapshot
17.14.2 Company Overview
17.14.3 Financial Analysis
17.14.3.1 Net Revenue, 2017-2021
17.14.3.2 Regional Revenue/Market Shares, 2021
17.14.3.3 Segmental Revenue/Market Shares, 2021
17.14.4 Product Benchmarking
17.14.5 Strategic Outlook
17.15 Pfizer CentreOne
17.15.1 Company Snapshot
17.15.2 Company Overview
17.15.3 Financial Analysis
17.15.3.1 Net Revenue, 2017-2021
17.15.3.2 Regional Revenue/Market Shares, 2021
17.15.3.3 Segmental Revenue/Market Shares, 2021
17.15.4 Product Benchmarking
17.15.5 Strategic Outlook
17.16 Recipharm AB
17.16.1 Company Snapshot
17.16.2 Company Overview
17.16.3 Product Benchmarking
17.16.4 Strategic Outlook
17.17 Siegfried Holding AG
17.17.1 Company Snapshot
17.17.2 Company Overview
17.17.3 Financial Analysis
17.17.3.1 Net Revenue, 2017-2021
17.17.3.2 Regional Revenue/Market Shares, 2021
17.17.3.3 Segmental Revenue/Market Shares, 2021
17.17.4 Product Benchmarking
17.17.5 Strategic Outlook
17.18 Nexus Pharmaceuticals, Inc.
17.18.1 Company Snapshot
17.18.2 Company Overview
17.18.3 Product Benchmarking
17.18.4 Strategic Outlook
17.19 Avara Pharmaceutical Services, Inc.
17.19.1 Company Snapshot
17.19.2 Company Overview
17.19.3 Product Benchmarking
17.20 Eli Lilly and Company
17.20.1 Company Snapshot
17.20.2 Company Overview
17.20.3 Financial Analysis
17.20.3.1 Net Revenue, 2017-2021
17.20.3.2 Regional Revenue/Market Shares, 2021
17.20.4 Product Benchmarking
17.20.5 Strategic Outlook
17.21 Adare Pharma Solutions
17.21.1 Company Snapshot
17.21.2 Company Overview
17.21.3 Product Benchmarking
17.21.4 Strategic Outlook
18 Conclusion and Recommendations
18.1 Concluding Remarks from Visiongain
18.2 Recommendations for Market Players
List of Tables
Table 1 CMO of Sterile Injectable Drugs Market Snapshot, 2022 & 2032 (US$ million, CAGR %)
Table 2 CMO of Sterile Injectable Drugs Market by Region, 2022-2032 (US$ mn, AGR (%), CAGR (%)): “V” Shaped Recovery Scenario
Table 3 CMO of Sterile Injectable Drugs Market by Region, 2022-2032 (US$ mn, AGR (%), CAGR (%)): “U” Shaped Recovery Scenario
Table 4 CMO of Sterile Injectable Drugs Market by Region, 2022-2032 (US$ mn, AGR (%), CAGR (%)): “W” Shaped Recovery Scenario
Table 5 CMO of Sterile Injectable Drugs Market by Region, 2022-2032 (US$ mn, AGR (%), CAGR (%)): “L” Shaped Recovery Scenario
Table 6 CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 7 CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 8 CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR%, CAGR %)
Table 9 CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 10 CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR%, CAGR %)
Table 11 CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR%, CAGR %)
Table 12 CMO of Sterile Injectable Drugs Market by Region, 2022-2032 (USD Mn, AGR (%), CAGR (%))
Table 13 North America CMO of Sterile Injectable Drugs Market Forecast by Country, 2022-2032 (US$ million, AGR%, CAGR %)
Table 14 North America CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 15 North America CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR%, CAGR%)
Table 16 North America CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR%, CAGR%)
Table 17 North America CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR%, CAGR%)
Table 18 North America CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR%, CAGR %)
Table 19 North America CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR%, CAGR %)
Table 20 U.S. CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR%)
Table 21 Canada CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR%)
Table 22 Europe CMO of Sterile Injectable Drugs Market Forecast by Country, 2022-2032 (US$ million, AGR%, CAGR %)
Table 23 Europe CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR%, CAGR%)
Table 24 Europe CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 25 Europe CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR%, CAGR %)
Table 26 Europe CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 27 Europe CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR%, CAGR %)
Table 28 Europe CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR%, CAGR %)
Table 29 Germany CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 30 U.K. CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 31 France CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 32 Italy CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 33 Spain CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR%)
Table 34 Russia CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 35 Rest of Europe CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 36 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Country, 2022-2032 (US$ million, AGR%, CAGR %)
Table 37 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 38 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 39 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR%, CAGR %)
Table 40 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 41 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR%, CAGR %)
Table 42 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR%, CAGR%)
Table 43 China CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 44 Japan CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR%)
Table 45 India CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR%)
Table 46 Australia CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR%)
Table 47 South-East Asia CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 48 Rest of Asia Pacific CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 49 Latin America CMO of Sterile Injectable Drugs Market Forecast by Country, 2022-2032 (US$ million, AGR%, CAGR %)
Table 50 Latin America CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR%, CAGR%)
Table 51 Latin America CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 52 Latin America CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR%, CAGR %)
Table 53 Latin America CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR%, CAGR%)
Table 54 Latin America CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR%, CAGR %)
Table 55 Latin America CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR%, CAGR %)
Table 56 Brazil CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 57 Mexico CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR%)
Table 58 Rest of Latin America CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR%)
Table 59 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Country, 2022-2032 (US$ million, AGR%, CAGR %)
Table 60 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR%, CAGR%)
Table 61 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR%, CAGR %)
Table 62 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR%, CAGR %)
Table 63 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR%, CAGR%)
Table 64 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR%, CAGR %)
Table 65 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR%, CAGR %)
Table 66 GCC CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR%)
Table 67 South Africa CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR %)
Table 68 Rest of Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR%, CAGR%)
Table 69 Company Ranking, 2021
Patheon, Inc. (Thermo Fischer)
Table 70 Strategic Outlook 2018-2022
Table 71 Aenova Group: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 72 Aenova Group: Product Benchmarking
Table 73
Aenova Group: Strategic Outlook
Table 74 Almac Group: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 75 Almac Group: Product Benchmarking
Table 76
Almac Group: Strategic Outlook
Table 77 Baxter International Inc.: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 78 Baxter International Inc.: Product Benchmarking
Table 79
Baxter International Inc. : Strategic Outlook
Table 80 Boehringer Ingelheim International GmbH: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 81 Boehringer Ingelheim International GmbH: Product Benchmarking
Table 82 Catalent, Inc. : Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 83 Catalent, Inc.: Product Benchmarking
Table 84
Catalent, Inc: Strategic Outlook
Table 85 CordenPharma: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 86 CordenPharma: Product Benchmarking
Table 87
CordenPharma: Strategic Outlook
Table 88 Evonik Industries AG: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 89 Evonik Industries AG: Product Benchmarking
Table 90
Evonik Industries AG: Strategic Outlook
Table 91 FAMAR Health Care Services: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 92 FAMAR Health Care Services: Product Benchmarking
Table 93
FAMAR Health Care Services: Strategic Outlook
Table 94 Fresenius Kabi Ag: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 95 Fresenius Kabi Ag: Product Benchmarking
Table 96
Fresenius Kabi Ag: Strategic Outlook
Table 97 Grifols SA: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 98 Grifols SA: Product Benchmarking
Table 99
Grifols SA: Strategic Outlook
Table 100 Hikma Pharmaceuticals PLC: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 101 Hikma Pharmaceuticals PLC: Product Benchmarking
Table 102
Hikma Pharmaceuticals PLC: Strategic Outlook
Table 103 Jubilant Pharmova Limited: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 104 Jubilant Pharmova Limited: Product Benchmarking
Table 105
Jubilant Pharmova Limited: Strategic Outlook
Table 106 Lonza: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 107 Lonza: Product Benchmarking
Table 108
Lonza: Strategic Outlook
Table 109 Patheon, Inc. (Thermo Fischer): Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 110 Patheon, Inc. (Thermo Fischer): Product Benchmarking
Table 111
Patheon, Inc. (Thermo Fischer): Strategic Outlook
Table 112 Pfizer CentreOne: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 113 Pfizer CentreOne: Product Benchmarking
Table 114
Pfizer CentreOne: Strategic Outlook
Table 115 Recipharm AB: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 116 Recipharm AB : Product Benchmarking
Table 117
Recipharm AB: Strategic Outlook
Table 118 Siegfrid Holding AG: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 119 Siegfried Holding AG: Product Benchmarking
Table 120
Siegfried Holding AG: Strategic Outlook
Table 121 Nexus Pharmaceuticals, Inc. Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 122 Nexcus Pharmaceuticals, Inc.: Product Benchmarking
Table 123
Nexus Pharmaceuticals, Inc.: Strategic Outlook
Table 124 Avara Pharmaceutical Services, Inc.: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 125 Avara Pharmaceutical Services, inc.: Product Benchmarking
Table 126 Patheon, Inc. (Thermo Fischer): Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 127 Eli Lilly and Company: Product Benchmarking
Table 128 Eli Lilly and Company : Strategic Outlook
Table 129 Adare Pharma Solutions: Key Details, (CEO, HQ, Revenue, Founded, No. of Employees, Company Type, Website, Business Segment)
Table 130 Adare Pharma Solutions: Product Benchmarking
Table 131
Adare Pharma Solutions: Strategic Outlook
List of Figures
Figure 1 CMO of Sterile Injectable Drugs Market Segmentation
Figure 2 CMO of Sterile Injectable Drugs Market Forecast by Region: Market Attractiveness Index
Figure 3 CMO of Sterile Injectable Drugs Market by Molecule Type: Market Attractiveness Index
Figure 4 CMO of Sterile Injectable Drugs Market by Type: Market Attractiveness Index
Figure 5 CMO of Sterile Injectable Drugs Market by Application: Market Attractiveness Index
Figure 6 CMO of Sterile Injectable Drugs Market by Container Type: Market Attractiveness Index
Figure 7 CMO of Sterile Injectable Drugs Market by Route of Administration: Market Attractiveness Index
Figure 8 CMO of Sterile Injectable Drugs Market by Service: Market Attractiveness Index
Figure 9 CMO of Sterile Injectable Drugs Market: Market Dynamics
Figure 10 Pharmaceutical R&D Expenditure in Europe, U.S., Japan & China, 2019 and 2020
Figure 11 CMO of Sterile Injectable Drugs Market by Region, 2022-2032 (US$ mn, AGR (%), CAGR (%)): “V” Shaped Recovery
Figure 12 CMO of Sterile Injectable Drugs Market by Region, 2022-2032 (US$ mn, AGR (%), CAGR (%)): “U” Shaped Recovery
Figure 13 CMO of Sterile Injectable Drugs Market by Region, 2022-2032 (US$ mn, AGR (%), CAGR (%)): “W” Shaped Recovery
Figure 14 CMO of Sterile Injectable Drugs Market by Region, 2022-2032 (US$ mn, AGR (%), CAGR (%)): “L” Shaped Recovery
Figure 15 CMO of Sterile Injectable Drugs Market: Porter’s Five Forces Analysis
Figure 16 CMO of Sterile Injectable Drugs Market: PEST Analysis
Figure 17 CMO of Sterile Injectable Drugs Market Forecast by Molecule Type 2022, 2027, 2032 (Revenue, CAGR %)
Figure 18 CMO of Sterile Injectable Drugs Market Share Forecast by Molecule Type, 2022, 2032 (%)
Figure 19 CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR %)
Figure 20 CMO of Sterile Injectable Drugs Market Share Forecast by Molecule Type, 2022, 2027, 2032 (%)
Figure 21 CMO of Sterile Injectable Drugs Market Forecast by Type 2022, 2027, 2032 (Revenue, CAGR %)
Figure 22 CMO of Sterile Injectable Drugs Market Share Forecast by Type, 2022, 2032 (%)
Figure 23 CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR %)
Figure 24 CMO of Sterile Injectable Drugs Market Share Forecast by Type, 2022, 2027, 2032 (%)
Figure 25 CMO of Sterile Injectable Drugs Market Forecast by Application 2022, 2027, 2032 (Revenue, CAGR %)
Figure 26 CMO of Sterile Injectable Drugs Market Share Forecast by Application, 2022, 2032 (%)
Figure 27 CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR %)
Figure 28 CMO of Sterile Injectable Drugs Market Share Forecast by Application, 2022, 2027, 2032 (%)
Figure 29 CMO of Sterile Injectable Drugs Market Forecast by Container Type 2022, 2027, 2032 (Revenue, CAGR %)
Figure 30 CMO of Sterile Injectable Drugs Market Share Forecast by Container Type, 2022, 2032 (%)
Figure 31 CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR %)
Figure 32 CMO of Sterile Injectable Drugs Market Share Forecast by Container Type, 2022, 2027, 2032 (%)
Figure 33 CMO of Sterile Injectable Drugs Market Forecast by Route of Administration 2022, 2027, 2032 (Revenue, CAGR %)
Figure 34 CMO of Sterile Injectable Drugs Market Share Forecast by Route of Administration, 2022, 2032 (%)
Figure 35 CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR %)
Figure 36 CMO of Sterile Injectable Drugs Market Share Forecast by Route of Administration, 2022, 2027, 2032 (%)
Figure 37 CMO of Sterile Injectable Drugs Market Forecast by Service 2022, 2027, 2032 (Revenue, CAGR %)
Figure 38 CMO of Sterile Injectable Drugs Market Share Forecast by Service, 2022, 2032 (%)
Figure 39 CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR %)
Figure 40 CMO of Sterile Injectable Drugs Market Share Forecast by Service, 2022, 2027, 2032 (%)
Figure 41 CMO of Sterile Injectable Drugs Market Share Forecast by Region 2022, 2027, 2032(%)
Figure 42 CMO of Sterile Injectable Drugs Market by Region, 2022-2032 (US$ mn, AGR (%), CAGR (%))
Figure 43 North America CMO of Sterile Injectable Drugs Market Attractiveness Index
Figure 44 North America CMO of Sterile Injectable Drugs Market by Region, 2022, 2027 & 2032 (US$ million)
Figure 45 North America CMO of Sterile Injectable Drugs Market Forecast by Country, 2022-2032 (US$ million, AGR %)
Figure 46 North America CMO of Sterile Injectable Drugs Market Share Forecast by Country, 2022 & 2032 (%)
Figure 47 North America CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR %)
Figure 48 North America CMO of Sterile Injectable Drugs Market Share Forecast by Molecule Type, 2022 & 2032 (%)
Figure 49 North America CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR %)
Figure 50 North America CMO of Sterile Injectable Drugs Market Share Forecast by Type, 2022 & 2032 (%)
Figure 51 North America CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR %)
Figure 52 North America CMO of Sterile Injectable Drugs Market Share Forecast by Application, 2022 & 2032 (%)
Figure 53 North America CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR %)
Figure 54 North America CMO of Sterile Injectable Drugs Market Share Forecast by Container Type, 2022 & 2032 (%)
Figure 55 North America CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR %)
Figure 56 North America CMO of Sterile Injectable Drugs Market Share Forecast by Route of Administration, 2022 & 2032 (%)
Figure 57 North America CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR %)
Figure 58 North America CMO of Sterile Injectable Drugs Market Share Forecast by Service, 2022 & 2032 (%)
Figure 59 U.S. CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 60 Canada CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 61 Europe CMO of Sterile Injectable Drugs Market Attractiveness Index
Figure 62 Europe CMO of Sterile Injectable Drugs Market by Region, 2022, 2027 & 2032 (US$ million)
Figure 63 Europe CMO of Sterile Injectable Drugs Market Forecast by Country, 2022-2032 (US$ million, AGR %)
Figure 64 Europe CMO of Sterile Injectable Drugs Market Share Forecast by Country, 2022 & 2032 (%)
Figure 65 Europe CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR %)
Figure 66 Europe CMO of Sterile Injectable Drugs Market Share Forecast by Molecule Type, 2022 & 2032 (%)
Figure 67 Europe CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR %)
Figure 68 Europe CMO of Sterile Injectable Drugs Market Share Forecast by Type, 2022 & 2032 (%)
Figure 69 Europe CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR %)
Figure 70 Europe CMO of Sterile Injectable Drugs Market Share Forecast by Application, 2022 & 2032 (%)
Figure 71 Europe CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR %)
Figure 72 Europe CMO of Sterile Injectable Drugs Market Share Forecast by Container Type, 2022 & 2032 (%)
Figure 73 Europe CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR %)
Figure 74 Europe CMO of Sterile Injectable Drugs Market Share Forecast by Route of Administration, 2022 & 2032 (%)
Figure 75 Europe CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR %)
Figure 76 Europe CMO of Sterile Injectable Drugs Market Share Forecast by Service, 2022 & 2032 (%)
Figure 77 Germany CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 78 U.K. CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 79 France CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 80 Italy CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 81 Spain CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 82 Russia CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 83 Rest of Europe CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 84 Asia Pacific CMO of Sterile Injectable Drugs Market Attractiveness Index
Figure 85 Asia Pacific CMO of Sterile Injectable Drugs Market by Region, 2022, 2027 & 2032 (US$ million)
Figure 86 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Country, 2022-2032 (US$ million, AGR %)
Figure 87 Asia Pacific CMO of Sterile Injectable Drugs Market Share Forecast by Country, 2022 & 2032 (%)
Figure 88 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR %)
Figure 89 Asia Pacific CMO of Sterile Injectable Drugs Market Share Forecast by Molecule Type, 2022 & 2032 (%)
Figure 90 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR %)
Figure 91 Asia Pacific CMO of Sterile Injectable Drugs Market Share Forecast by Type, 2022 & 2032 (%)
Figure 92 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR %)
Figure 93 Asia Pacific CMO of Sterile Injectable Drugs Market Share Forecast by Type, 2022 & 2032 (%)
Figure 94 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR %)
Figure 95 Asia Pacific CMO of Sterile Injectable Drugs Market Share Forecast by Container Type, 2022 & 2032 (%)
Figure 96 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR %)
Figure 97 Asia Pacific CMO of Sterile Injectable Drugs Market Share Forecast by Route of Administration, 2022 & 2032 (%)
Figure 98 Asia Pacific CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR %)
Figure 99 Asia Pacific CMO of Sterile Injectable Drugs Market Share Forecast by Service, 2022 & 2032 (%)
Figure 100 China CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 101 Japan CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 102 India CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 103 Australia CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 104 South-East Asia CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 105 Rest of Asia Pacific CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 106 Latin America CMO of Sterile Injectable Drugs Market Attractiveness Index
Figure 107 Latin America CMO of Sterile Injectable Drugs Market by Region, 2022, 2027 & 2032 (US$ million)
Figure 108 Latin America CMO of Sterile Injectable Drugs Market Forecast by Country, 2022-2032 (US$ million, AGR %)
Figure 109 Latin America CMO of Sterile Injectable Drugs Market Share Forecast by Country, 2022 & 2032 (%)
Figure 110 Latin America CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR %)
Figure 111 Latin America CMO of Sterile Injectable Drugs Market Share Forecast by Molecule Type, 2022 & 2032 (%)
Figure 112 Latin America CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR %)
Figure 113 Latin America CMO of Sterile Injectable Drugs Market Share Forecast by Type, 2022 & 2032 (%)
Figure 114 Latin America CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR %)
Figure 115 Latin America CMO of Sterile Injectable Drugs Market Share Forecast by Application, 2022 & 2032 (%)
Figure 116 Latin America CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR %)
Figure 117 Latin America CMO of Sterile Injectable Drugs Market Share Forecast by Container Type, 2022 & 2032 (%)
Figure 118 Latin America CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR %)
Figure 119 Latin America CMO of Sterile Injectable Drugs Market Share Forecast by Route of Administration, 2022 & 2032 (%)
Figure 120 Latin America CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR %)
Figure 121 Latin America CMO of Sterile Injectable Drugs Market Share Forecast by Service, 2022 & 2032 (%)
Figure 122 Brazil CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 123 Mexico CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 124 Rest of Latin America CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 125 Middle East & Africa CMO of Sterile Injectable Drugs Market Attractiveness Index
Figure 126 Middle East & Africa CMO of Sterile Injectable Drugs Market by Region, 2022, 2027 & 2032 (US$ million)
Figure 127 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Country, 2022-2032 (US$ million, AGR %)
Figure 128 Middle East & Africa CMO of Sterile Injectable Drugs Market Share Forecast by Country, 2022 & 2032 (%)
Figure 129 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Molecule Type, 2022-2032 (US$ million, AGR %)
Figure 130 Middle East & Africa CMO of Sterile Injectable Drugs Market Share Forecast by Molecule Type, 2022 & 2032 (%)
Figure 131 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Type, 2022-2032 (US$ million, AGR %)
Figure 132 Middle East & Africa CMO of Sterile Injectable Drugs Market Share Forecast by Type, 2022 & 2032 (%)
Figure 133 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Application, 2022-2032 (US$ million, AGR %)
Figure 134 Middle East & Africa CMO of Sterile Injectable Drugs Market Share Forecast by Application, 2022 & 2032 (%)
Figure 135 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Container Type, 2022-2032 (US$ million, AGR %)
Figure 136 Middle East & Africa CMO of Sterile Injectable Drugs Market Share Forecast by Container Type, 2022 & 2032 (%)
Figure 137 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Route of Administration, 2022-2032 (US$ million, AGR %)
Figure 138 Middle East & Africa CMO of Sterile Injectable Drugs Market Share Forecast by Route of Administration, 2022 & 2032 (%)
Figure 139 Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast by Service, 2022-2032 (US$ million, AGR %)
Figure 140 Middle East & Africa CMO of Sterile Injectable Drugs Market Share Forecast by Service, 2022 & 2032 (%)
Figure 141 GCC CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 142 South Africa CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
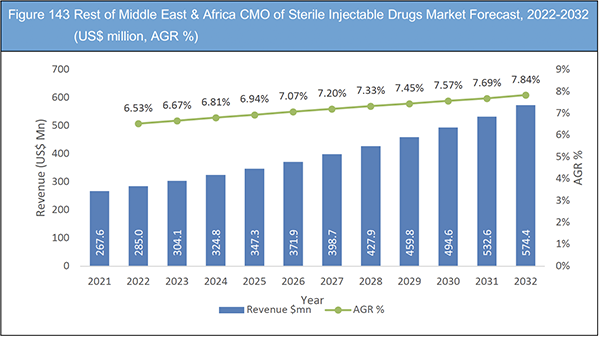
Figure 143 Rest of Middle East & Africa CMO of Sterile Injectable Drugs Market Forecast, 2022-2032 (US$ million, AGR %)
Figure 144 Baxter International Inc. : Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 145 Baxter International Inc. : Regional Revenue Shares, 2021
Figure 146 Boehringer Ingelheim International GmbH: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 147 Boehringer Ingelheim International GmbH: Regional Revenue Shares, 2021
Figure 148 Boehringer Ingelheim International GmbH : Segmental Revenue Shares, 2021
Figure 149 Catalent, Inc.: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 150 Catalent, Inc.: Regional Revenue Shares, 2021
Figure 151 Catalent, Inc.: Segmental Revenue Shares, 2021
Figure 152 Evonik Industries AG: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 153 Evonik Industries AG : Regional Revenue Shares, 2021
Figure 154 Evonik Industries AG: Segmental Revenue Shares, 2021
Figure 155 Fresenius Kabi Ag: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 156 Fresenius Kabi Ag: Regional Revenue Shares, 2021
Figure 157 Fresenius Kabi Ag: Segmental Revenue Shares, 2021
Figure 158 Grifols SA: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 159 Grifols SA: Regional Revenue Shares, 2021
Figure 160 Grifols SA : Segmental Revenue Shares, 2021
Figure 161 Hikma Pharmaceuticals PLC: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 162 Hikma Pharmaceuticals PLC: Regional Revenue Shares, 2021
Figure 163 Hikma Pharmaceuticals PLC: Segmental Revenue Shares, 2021
Figure 164 Jubilant Pharmova Limited: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 165 Jubilant Pharmova Limited. : Regional Revenue Shares, 2021
Figure 166 Jubilant Pharmova Limited : Segmental Revenue Shares, 2021
Figure 167 Lonza: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 168 Lonza: Regional Revenue Shares, 2021
Figure 169 Lonza: Segmental Revenue Shares, 2021
Figure 170 Patheon, Inc. (Thermo Fischer): Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 171 Patheon, Inc. (thermo Fischer): Regional Revenue Shares, 2021
Figure 172 Patheon, Inc. (Thermo Fischer): Segmental Revenue Shares, 2021
Figure 173 Pfizer CentreOne: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 174 Pfizer CentreOne : Regional Revenue Shares, 2021
Figure 175 Pfizer CentreOne : Segmental Revenue Shares, 2021
Figure 176 Siegfried Holding AG: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 177 Siegfried Holding AG: Regional Revenue Shares, 2021
Figure 178 Siegfried Holding AG: Segmental Revenue Shares, 2021
Figure 179 Eli Lilly and Company: Net Revenue, 2017-2021 (US$ million, AGR %)
Figure 180 Eli Lilly and Company: Regional Revenue Shares, 2021
List of Companies Profiled in the Report
Adare Pharma Solutions
Aenova Group
Almac Group
Avara Pharmaceutical Services, Inc.
Baxter International Inc.
Boehringer Ingelheim International GmbH
Catalent, Inc.
CordenPharma
Eli Lilly and Company
Evonik Industries AG
F. Hoffmann-La Roche Ltd
FAMAR Health Care Services
Fresenius Kabi Ag
Grifols SA
Hikma Pharmaceuticals PLC
Jubilant Pharmova Limited
Lonza
Nexus Pharmaceuticals, Inc.
Patheon, Inc. (Thermo Fischer)
Pfizer CentreOne
Recipharm AB
Siegfried Holding AG
List of Other Companies Mentioned in the Report
Acucela
Autolus
AveXis
BioCancell
Biomay
Biomiga
BioReliance
Biotec Services International
Celladon
Cellectis
Cellular Biomedicine Group
Delphi Genetics
Desktop Genetics
DNAtrix
Elixirgen Scientific
Epeius Biotechnologies
EUFETS
Eurofins Genomics
GEG Tech
Genable Technologies
Immune Application
Immune Design
ImmunoGenes
Inbiomed
VIVEbiotech
Waisman Biomanufacturing
Xpress Biologics
List of Associations Mentioned in the Report
Association of British Pharmaceutical Industry (ABPI)
Central Drugs Standard Control Organization (CDSCO)
Department of Health and Family Welfare
Development and Reform Commission (NDRC)
European Commission
European Medicines Agency (EMA)
Food and Drug Administration (US FDA)
Indian Drug Manufacturer's Association (IDMA)
International Society of Pharmaceutical Engineering (IPSE)
Korea Food and Drug Administration (KFDA)
Korea Pharmaceutical Manufacturer's Association (KPMA)
Medicines and Healthcare Products Regulatory Agency (MHRA)
Medicines Manufacturing Industry Partnership (MMIP)
Ministry of Health (MOH)
Ministry of Health, Labor and Welfare (MHLW)
Ministry of Industry and Information Application (MIIT)
Pharmaceutical and Medical Devices Agency (PMDA)
World Health Organization (WHO)





























