Industries > Pharma > Clinical Trial Supply and Logistics Market for Pharma 2018-2028
Clinical Trial Supply and Logistics Market for Pharma 2018-2028
Clinical Trial Manufacturing, Clinical Trial Logistics and Distributions, Clinical Trial Supply Chain Management, Clinical Trial Packaging, Clinical Trial Cold Chain Logistics
The Global Clinical Trials Supply and Logistics Market is estimated at $15.77bn in 2017 and is expected to grow at a CAGR of 6.9% from 2017-2022. In 2017, this market was dominated by the Clinical Trial Manufacturing submarket which held 66.30% of the overall market.
How this report will benefit you
Read on to discover how you can exploit the future business opportunities emerging in this sector.
In this brand new 202-page report you will receive 117 charts– all unavailable elsewhere.
The 202-page report provides clear detailed insight into the Global Clinical Trial Supply and Logistics Market. Discover the key drivers and challenges affecting the market.
By ordering and reading our brand-new report today you stay better informed and ready to act.
Report Scope
• Revenue and growth forecasts from 2018 to 2028 for the global clinical trial supply and logistics market. This report also discusses the Drivers and Restraints of the global clinical trial supply and logistics market.
• Revenue and growth forecasts from 2018 to 2028 for the leading submarkets of the global clinical trial supply and logistics market:
– Manufacturing – also with sub forecasting for packaging and for other production
– Logistics and distribution – also with sub forecasts for cold chain logistics and for other services
– Supply chain management
This report discusses the Drivers and Restraints of each submarket.
• Revenue and growth forecasts from 2018 to 2028 for the leading national markets:
– United States
– Japan
– Germany
– France
– United Kingdom
– Italy
– Spain
– Brazil
– Russia
– China
– India
– Rest of the World
• This report profiles the leading companies offering clinical trial supply and logistics services to the pharmaceutical industry:
– World Courier
– DHL
– Marken
– FedEx
– Movianto
– Catalent
– Patheon
– Almac Group
– Parexel International
– Fisher Clinical Services
– Packaging Coordinators Inc.
• This report provides qualitative analysis of the clinical trial supply and logistics market. This report discusses the Strengths, Weaknesses, Opportunities and Threats of the clinical trial supply and logistics market. Social, Technological, Economic and Political factors that influence this market are also discussed.
• This report discusses trends in the clinical trial supply and logistics market, clinical trial manufacturing market, comparator sourcing, clinical trial packaging, clinical trial supply chain management.
• This report discusses the regulatory outlook of the clinical trial supply and logistics industry, outlook for cold chain logistics in the clinical trial sector, as well as regulatory aspects of cold chain distribution for clinical trial materials
• This report also provides a full transcript of an interview conducted by Visiongain with Marken.
Visiongain’s study is intended for anyone requiring commercial analyses for the global clinical trial supply and logistics market. You find data, trends and predictions.
Buy our report today Clinical Trial Supply and Logistics Market for Pharma 2018-2028: Clinical Trial Manufacturing, Clinical Trial Logistics and Distributions, Clinical Trial Supply Chain Management, Clinical Trial Packaging, Clinical Trial Cold Chain Logistics.
Visiongain is a trading partner with the US Federal Government
CCR Ref number: KD4R6
1. Report Overview
1.1 Overview of the World Market for Clinical Trial Supply and Logistics
1.2 Benefits of This Report
1.3 How This Report Delivers
1.4 Main Questions Answered by This Analysis
1.5 Who Is This Study For?
1.6 Methods of Research and Analysis
1.7 Frequently Asked Questions (FAQs)
1.8 Some Related Reports
1.9 About Visiongain
2. Defining the Clinical Trial Supply and Logistics Market
2.1 An Introduction to the Clinical Trial Supply and Logistics Market
2.2 Clinical Trials are Becoming Increasingly Complex
2.3 Clinical Trial Globalisation: Access to New Markets and New Patients
2.4 Outsourcing Clinical Trial Supply and Logistics
2.5 What Services Are Most Commonly Outsourced?
2.6 Which Companies Participate in the Clinical Trial Supply and Logistics Market?
2.7 Clinical Trial Supply and Logistics: Trends and Future Outlook
3. Clinical Trial Supply and Logistics: World Market Outlook and Forecasting 2017-2028
3.1 The World Clinical Trial Supply and Logistics Market 2017
3.1.1 Which Were the Leading Market Sectors in 2017?
3.2 The World Clinical Trial Supply and Logistics Market Forecast 2017-2028
3.2.1 What Will Drive Market Growth to 2028?
3.2.2 Clinical Trial Supply and Logistics: Market Restraints 2018-2028
3.3 Clinical Trial Manufacturing Submarket 2017-2028
3.3.1 Outlook and Forecast for the Clinical Trial Manufacturing Submarket 2017-2028
3.3.2 Complex Manufacturing and Packaging to Drive Growth 2017-2028
3.3.3 Clinical Trial Packaging Submarket Forecast 2017-2028
3.4 Clinical Trial Logistics and Distribution Submarket 2017-2028
3.4.1 Clinical Trial Logistics and Distribution Submarket Forecast 2017-2028
3.4.2 Clinical Trial Logistics and Distribution: Drivers and Restraints 2017-2028
3.4.3 Clinical Trial Cold Chain Distribution Submarket Forecast 2017-2028
3.5 Clinical Trial Supply Chain Management Submarket 2017-2028
3.5.1 Clinical Trial Supply Chain Management Submarket Forecast 2017-2028
3.5.2 Drivers and Restraints for the Clinical Trial Supply Chain Management Submarket 2017-2028
4. Leading National Markets for Clinical Trial Supply and Logistics 2017-2028
4.1 Defining National Submarkets for Clinical Trial Supply and Logistics
4.2 Leading National Submarkets for Clinical Trial Supply and Logistics 2017
4.2.1 How Will These Leading Submarkets Grow 2017-2028?
4.3 The US: Clinical Trial Supply and Logistics Market, 2017-2028
4.3.1 How Will Demand for Clinical Trial Logistics Grow in the US to 2028?
4.4 EU5 Markets for Clinical Trial Supply and Logistics, 2017-2028
4.4.1 The Clinical Trial Supply and Logistics Market for Germany: 2017-2028
4.4.2 France: Clinical Trial Supply and Logistics Market, 2017-2028
4.4.3 The UK: Clinical Trial Supply and Logistics Market, 2017-2028
4.4.4 Italy: Clinical Trial Supply and Logistics Market, 2017-2028
4.4.5 Spain: Clinical Trial Supply and Logistics Market, 2017-2028
4.5 Japan: A New and Growing Market for Outsourced Logistics
4.5.1 Japanese Clinical Trial Supply and Logistics Submarket Forecast 2017-2028
4.6 Clinical Trials Increasing Demand in Emerging National Markets
4.7 China: Clinical Trial Supply and Logistics Market, 2017-2028
4.7.1 Foreign Investment in Chinese Clinical Trial Logistics
4.7.2 Strong Growth for Clinical Trial Supply and Logistics in China 2017-2028
4.8 India: An Emerging Destination for Outsourced Clinical Trials
4.8.2 India: Clinical Trial Supply and Logistics Market, 2017-2028
4.9 The Latin American Clinical Trial Supply and Logistics Market
4.9.1 Challenging Distribution Conditions
4.9.2 Brazil: Clinical Trial Supply and Logistics Market, 2017-2028
4.10 Russia: Clinical Trial Supply and Logistics Market, 2017-2028
4.10.1 Russia Benefiting from Close Proximity to European Union
4.10.2 Outsourcing Will Grow Strongly in Russia
5. Clinical Trial Supply and Logistics: Regulatory Outlook 2017-2028
5.1 How do Regulatory Standards Affect Clinical Trial Supply?
5.1.1 Good Practices (GxP) in Clinical Trial Distribution
5.1.1.1 Good Manufacturing Practice (GMP)
5.1.1.2 Good Distribution Practice (GDP)
5.1.2 Import Regulations Complicate International Manufacturing and Trials
5.2 Clinical Trial Supply Regulations in the US 2017
5.2.1 Importing Drugs into the US: A Reinterpretation of the Rules
5.3 Clinical Trial Regulations in the EU 2017
5.3.1 Manufacturing Requirements for Clinical Trial Materials in the EU
5.3.2 The European Commission Releases New GDP Guidelines 2013
5.4 GMP and Importation Regulations for Pharmaceuticals in Japan 2017
5.5 Clinical Trial Supply Chain Regulations in Emerging Markets
5.5.1 China: Tougher Regulations for Pharmaceutical Companies and Service Providers
5.5.1.1 Improving Supply Chain Security in China 2017
5.5.2 India: Drug Manufacturing and Imports for Clinical Trials
5.5.2.1 The CDSCO Drafts New GDP Guidelines
5.5.3 Brazil: Slow Regulatory Approval for Clinical Trials
5.5.4 Recent Updates to Pharmaceutical Regulation in Russia
6. Clinical Trial Supply and Logistics: Industry Trends 2017-2028
6.1 Clinical Trial Supply and Logistics Market: Strengths and Weaknesses 2017
6.2 Clinical Trial Supply and Logistics Market: Opportunities and Threats 2017-2028
6.3 Clinical Trial Supply and Logistics Market: STEP Analysis 2017-2028
6.3.1 Social Factors
6.3.2 How Will Novel Technologies Affect the Market to 2028?
6.3.3 Economic Pressure: Demands for Lower Cost Trials and Logistics
6.3.4 Political Issues
6.4 Securing the Clinical Trial Supply Chain 2017-2028
6.4.1 How Will Technology Improve Supply Chain Security?
6.5 How Will Clinical Trial Globalisation Impact Supply and Logistics?
6.6 Trends in Clinical Trial Manufacturing 2017-2028
6.6.1 The Challenge of Comparator Sourcing for Clinical Trials
6.6.1.1 Comparators Are a Source of Risk for Supply Chain Security
6.6.1.2 Regulators and Payers Will Increasingly Demand Comparative Effectiveness Research
6.6.2 Trends in Packaging for Clinical Trials 2017-2028
6.7 Clinical Trial Supply Chain Management Trends to 2028
6.7.1 Forecasting and Modelling to Ensure Sufficient Supply
6.7.2 IT Systems Improve Clinical Trial Planning: IVRS and IWRS
6.7.3 Home-Based Trial Participation: Opportunities and Challenges for Logistics
6.7.4 How Will Greater Use of Adaptive Clinical Trials Impact Trial Logistics?
6.8 Outsourcing Clinical Trial Supply and Logistics
6.8.1 Managing Multiple Supplier Networks
7. The Rise of Cold Chain Logistics in Clinical Trials 2017-2028
7.1 An Introduction to Cold Chain Supply and Distribution
7.1.1 The Importance of Cold Chain for Clinical Trials
7.1.2 Maintaining the Cold Chain
7.2 The Regulatory Outlook for Cold Chain Logistics
7.2.1 International Air Transport Regulations and Guidance: The Temperature Sensitive Label
7.3 Cold Chain Will Account for an Increasing Proportion of Clinical Trial Logistics 2017-2028
7.3.1 Biologic Drug Development Will Stimulate Demand
7.3.1.1 Emerging Trends in Biological Drug Development
7.3.2 Pharma Cold Chain Logistics in Emerging Markets
7.4 The Role of Technology in Clinical Cold Chain Logistics
7.4.1 Active and Passive Temperature Control
7.4.2 There Are Many Ways to Maintain Temperature in the Cold Chain
7.4.2.1 Developments in Phase Change Materials
7.4.2.2 The Benefits of Liquid Nitrogen for Sample Transportation
7.4.3 Advances in RFID for Temperature Monitoring
7.4.3.1 Will Uptake for RFID Grow Among Biopharma Companies to 2028?
7.4.4 Future Technologies for Cold Chain Supply Management
7.4.4.1 Demand for Reusable Cold Chain Packaging
8. Clinical Trial Supply and Logistics Market: Leading Companies
8.1 Third Party Logistics Providers: Leading Clinical Distributors
8.1.1 World Courier: A Pharmaceutical Supply Specialist
8.1.1.1 Distribution Services for Pharma, CROs and Central Laboratories
8.1.1.2 World Courier Is the Clinical Trial Distribution Market Leader
8.1.1.3 World Courier Service Expansion
8.1.2 DHL: A Global Leader in Supply Chain Logistics
8.1.2.1 Life Sciences: A Fast-Growing Source of Revenue for DHL Supply Chain
8.1.2.2 DHL: Services for Healthcare and Clinical Trial Clients
8.1.3 Marken: A leader in Direct to Patient Services
8.1.3.1 What Services Does Marken Offer Clinical Trial Sponsors?
8.1.3.2 Marken: Challenging Market Conditions 2010-2015
8.1.3.3 Marken Continues to Expand Worldwide
8.1.3.4 Expertise in Biological Sample Management
8.1.4 Other Leading and Emerging Players in the Market 2017
8.1.4.1 FedEx
8.1.4.2 Movianto - A European Healthcare Specialist
8.2 Clinical Trial Manufacturing: Submarket Leaders
8.2.1 Catalent Pharma Solutions
8.2.1.1 Clinical Trial Manufacturing and Supply Services
8.2.1.2 Strong Growth for Clinical Manufacturing Revenue
8.2.1.3 Catalent Expanding its Clinical Supply Services
8.2.2 Patheon N.V.
8.2.2.1 Patheon – Global leader in Pharmaceutical Development Services
8.2.3 Almac Group: Manufacturing, Packaging and Logistics Services
8.2.3.1 Clinical Trial Supply and Management Solutions
8.2.3.2 Almac Opens New Facilities to Expand its Clinical Trial Expertise
8.3 The Role of CROs in the Clinical Trial Supply and Logistics Market
8.3.1 PAREXEL International Corp.
8.3.1.1 Parexel’s Clinical Trial Supply and Logistics Services
8.3.1.2 Parexel Expands Worldwide
8.4 Other Leading Companies in the Clinical Trial Supply and Logistics Market 2017
8.4.1 Fisher Clinical Services (FCS)
8.4.1.1 FCS Offers Manufacturing, Packaging, Storage and Distribution Services
8.4.1.2 Fisher BioServices: Clinical Logistics for the Cell Therapy Industry
8.4.2 Packaging Coordinators Inc. (PCI)
9. Research Interviews from Our Survey
9.1 Marken
9.1.1 Marken
9.1.2 Major Changes in The Clinical Trial Supply and Logistics Market
10. Conclusions of Our Study
10.1 The Clinical Trial Supply and Logistics Market in 2017
10.2 Outlook for the Market, 2017-2028
10.3 Growing Demand for Clinical Logistics in Emerging Countries
10.4 Technology and Innovation to Drive Outsourcing
Appendices
Associated Visiongain Reports
Visiongain Report Sales Order Form
About Visiongain
Report Evaluation Form
List of Tables
Table 1.1 Currency Exchange Rate to $US, 2017
Table 2.1 Clinical Trials Registered by Location, December 2017
Table 3.1 R&D Outsourcing Market: Submarket Size ($bn) and Market Share (%) by Sector, 2017
Table 3.2 Clinical Trial Supply and Logistics Market: Submarket Size ($bn)and Market Share (%) by Sector, 2017
Table 3.3 Clinical Trial Supply and Logistics Market Revenue Forecast by Sector: Revenue ($bn), AGR (%) and CAGR (%), 2017-2022
Table 3.4 Clinical Trial Supply and Logistics Market Revenue Forecasts by Sector: Revenue ($bn), AGR (%) and CAGR (%), 2022-2028
Table 3.5 Clinical Trial Supply and Logistics Market: Submarket Shares (%), 2017-2028
Table 3.6 Clinical Trial Manufacturing Submarket: Submarket Size ($bn) and Submarket Share (%) by Sector, 2017
Table 3.7 Clinical Trial Manufacturing Market Revenue Forecast by Submarket: Revenue ($bn), AGR (%) and CAGR (%), 2017-2022
Table 3.8 Clinical Trial Manufacturing Market Revenue Forecast by Submarket: Revenue ($bn), AGR (%) and CAGR (%), 2022-2028
Table 3.9 Clinical Trial Packaging Submarket: Revenue Forecast ($bn), AGR (%) and CAGR (%), 2017-2028
Table 3.10 Clinical Trial Logistics and Distribution Market: Submarket Size ($bn) and Submarket Share (%) by Sector, 2017
Table 3.11 Clinical Trial Logistics and Distribution Market Revenue Forecast by Submarket: Revenue ($bn), AGR (%) and CAGR (%), 2017-2022
Table 3.12 Clinical Trial Logistics and Distribution Market Revenue Forecast by Submarket: Revenue ($bn), AGR (%) and CAGR (%), 2022-2028
Table 3.13 Clinical Trial Cold Chain Logistics Submarket: Revenue ($bn), AGR (%) and CAGR (%), 2017-2028
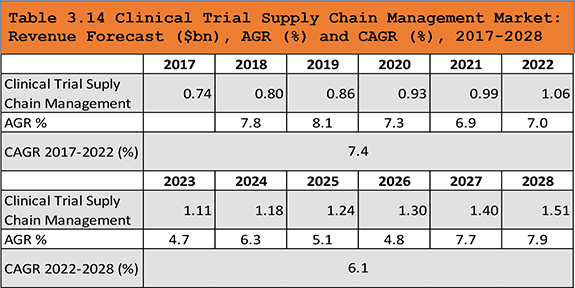
Table 3.14 Clinical Trial Supply Chain Management Market: Revenue Forecast ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.1 Clinical Trial Supply and Logistics Market: Market Size ($bn) and Market Shares (%) by Leading Country and Region, 2017
Table 4.2 Clinical Trial Supply and Logistics Market: Country Submarket Revenue Forecast ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.3 Clinical Trial Supply and Logistics Market: Leading National Market Share (%), 2017, 2022 and 2028
Table 4.4 US Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Table 4.5 EU5 Clinical Trial Supply and Logistics: Submarket Revenue Forecasts ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.6 The EU5 Clinical Trial Supply and Logistics Submarket: Market Size ($bn) and Market Shares (%), 2017
Table 4.7 Germany: Clinical Trial Supply and Logistics Market: Revenue ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.8 France Clinical Trial Supply and Logistics Submarket: Revenue ($bn), AGR (%) and CAGR (%),2017-2028
Table 4.9 The UK: Clinical Trial Supply and Logistics Market: Revenue ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.10 Italy: Clinical Trial Supply and Logistics Market: Revenue ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.11 Spain: Clinical Trial Supply and Logistics Market: Revenue ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.12 Japan: Clinical Trial Supply and Logistics Market: Revenue Forecast ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.13 China: Clinical Trial Supply and Logistics: Revenue Forecast ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.14 India: Clinical Trial Supply and Logistics: Revenue Forecast ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.15 Brazil: Clinical Trial Supply and Logistics Market: Revenue Forecast ($bn), AGR (%) and CAGR (%), 2017-2028
Table 4.16 Russia: Clinical Trial Supply and Logistics: Revenue Forecast ($bn), AGR (%) and CAGR (%), 2017-2028
Table 5.1 ICH Manufacturing Guidelines, 2001-2012
Table 6.1 Clinical Trial Supply and Logistics Market: Strengths and Weaknesses, 2017-2028
Table 6.2 Clinical Trial Supply and Logistics Market: Opportunities and Threats, 2017-2028
Table 6.3 Clinical Trial Supply and Logistics Market: STEP Analysis, 2017-2028
Table 6.4 Comparative Costs of Orphan and Non-Orphan Clinical Trials, 2015
Table 6.5 TransCelerate BioPharma Member Companies, 2017
Table 6.6 Benefits of Advanced Forecasting Tools for Clinical Trial Logistics
Table 6.7 Selected Clinical Trial Supply Forecasting Tools Available, 2016
Table 6.8 Selected IVRS/IWRS Tools Available for Clinical Trials, 2017
Table 6.9 Home-Based Clinical Trial Distribution: Advantages and Disadvantages, 2017
Table 6.10 Outsourcing Clinical Trial Supply and Logistics: Advantages and Disadvantages, 2017
Table 7.1 Top Ten Prescription Drugs: Overall Biologics Share, 2015 and 2016
Table 7.2 Leading Providers of RFID Tags for the Pharmaceutical Market, 2017
Table 8.1 AmerisourceBergen: Other Service Revenue ($bn), 2015-2017
Table 8.2 DHL Supply Chain: Revenue ($m) by Sector, 2016
Table 8.3 DHL Life Sciences and Healthcare: Revenue ($m) and AGR (%), 2014-2016
Table 8.4 Marken Historical Revenue: Revenue ($m), Annual Growth (%),2015-2017
Table 8.6 Catalent Clinical Supply Services: Revenue ($m), 2015-2017
Table 8.7 Patheon Pharmaceutical Development Projects (%) by Phase, 2016
Table 8.8 Patheon Pharmaceutical Development Services: Revenue ($bn), 2014-2016
Table 8.9 Almac Group: Revenue ($m), 2014-2016
Table 8.10 Parexel: Clinical Research Services Revenue ($m), 2012-2016
Table 10.1 Clinical Trial Supply and Logistics Market: Revenue ($bn) and Market Share (%) by Sector, 2017, 2022 and 2028
Table 10.2 Clinical Trial Supply and Logistics Market: Revenue ($bn) and Market Share (%) by Region in 2017, 2022 and 2028
List of Figures
Figure 2.1 Sectors and Services in the Clinical Trial Supply and Logistics Market, 2018
Figure 2.2 Clinical Trials Registered by Location, 2017
Figure 2.3 Trends Affecting the Clinical Trial Supply and Logistics Market, 2018-2028
Figure 3.1 Pharma R&D Outsourcing Market: Market Share (%) by Sector, 2017
Figure 3.2 Clinical Trial Supply and Logistics Market: Market Share (%) by Sector, 2017
Figure 3.3 Clinical Trial Supply and Logistics Market Forecast ($bn), 2017-2028
Figure 3.4 Clinical Trial Supply and Logistics Market: Submarket Shares (%), 2022
Figure 3.5 Clinical Trial Supply and Logistics Market: Submarket Shares (%), 2028
Figure 3.6 Clinical Trial Supply and Logistics: Market Drivers, 2017-2028
Figure 3.7 Clinical Trial Supply and Logistics: Market Restraints, 2017-2028
Figure 3.8 Clinical Trial Manufacturing Market: Sector Shares (%), 2017
Figure 3.9 Clinical Trial Manufacturing Market Forecast ($bn), 2017-2028
Figure 3.10 Clinical Trial Manufacturing: Drivers and Restraints, 2017-2028
Figure 3.11 Clinical Trial Packaging Submarket Forecast ($bn), 2017-2028
Figure 3.12 Clinical Trial Logistics and Distribution Market: Sector Shares (%), 2017
Figure 3.13 Clinical Trial Logistics and Distribution Market Forecast ($bn), 2017-2028
Figure 3.14 Clinical Trial Logistics and Distribution: Drivers and Restraints, 2017-2028
Figure 3.15 Clinical Trial Cold Chain Logistics Submarket Forecast ($bn), 2017-2028
Figure 3.16 Clinical Trial Supply Chain Management Market Forecast ($bn), 2017-2028
Figure 3.17 Clinical Trial Supply Chain Management: Drivers and Restraints, 2017-2028
Figure 4.1 Clinical Trial Supply and Logistics Market: Market Share (%) by Country and Region, 2017
Figure 4.2 Clinical Trial Supply and Logistics Market: Market Share (%) by Region, 2017, 2022 and 2028
Figure 4.3 US Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.4 US Clinical Trial Supply and Logistics Market Share (%), 2017, 2022 and 2028
Figure 4.5 US Clinical Trial Supply and Logistics: Drivers and Restraints, 2017-2028
Figure 4.6 EU5 Clinical Trial Supply and Logistics: Overall Revenue ($bn), 2017-2028
Figure 4.7 EU5 Clinical Trial Supply and Logistics Market Share (%), 2017, 2022 and 2028
Figure 4.8 EU5 Clinical Trial Supply and Logistics: Drivers and Restraints, 2017-2028
Figure 4.9 The EU5 Clinical Trial Supply and Logistics Submarket Shares (%), 2017
Figure 4.10 Germany: Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.11 France: Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.12 The UK: Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.13 Italy: Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.14 Spain: Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.15 Japan: Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.16 Japanese Clinical Trial Supply and Logistics Market Share (%) in 2017, 2022 and 2028
Figure 4.17 Japanese Clinical Trial Supply and Logistics: Drivers and Restraints, 2017-2028
Figure 4.18 China: Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.19 Chinese Clinical Trial Supply and Logistics Market Share (%), 2017, 2022 and 2028
Figure 4.20 Chinese Clinical Trial Supply and Logistics: Drivers and Restraints, 2017-2028
Figure 4.21 India: Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.22 Indian Clinical Trial Supply and Logistics Market Share (%), 2017, 2022 and 2028
Figure 4.23 Indian Clinical Trial Supply and Logistics: Drivers and Restraints, 2017-2028
Figure 4.24 Brazil: Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.25 Brazilian Clinical Trial Supply and Logistics Market Share (%), 2017, 2022 and 2028
Figure 4.26 Brazilian Clinical Trial Supply and Logistics: Drivers and Restraints, 2017-2028
Figure 4.27 Russia: Clinical Trial Supply and Logistics Market Revenue ($bn), 2017-2028
Figure 4.28 Russia: Clinical Trial Supply and Logistics Market Share (%), 2017, 2022 and 2028
Figure 4.29 Russian Clinical Trial Supply and Logistics: Drivers and Restraints, 2017-2028
Figure 6.1 Players in the Clinical Trial Supply Chain, 2017
Figure 8.1 AmerisourceBergen Other Service: Revenue ($bn), 2017
Figure 8.2 DHL Supply Chain: Revenue Share (%) by Sector, 2016
Figure 8.3 Marken: Revenue ($m), 2015-2017
Figure 8.4 Catalent: Clinical Manufacturing Space by Region (%), 2017
Figure 8.5 Catalent Clinical Supply Services: Revenue ($m), 2015-2017
Figure 8.6 Patheon Pharmaceutical Development Projects (%) by Phase, 2016
Figure 8.7 Patheon Pharmaceutical Development Services: Revenue ($bn), 2014-2016
Figure 8.8 Almac Group: Revenue ($m), 2014-2016
Figure 8.9 Parexel: Clinical Research Services Revenue ($m), 2012-2016
Figure 10.1 Clinical Trial Supply and Logistics Market: Revenue ($bn) by Sector, 2017, 2022 and 2028
Figure 10.2 Clinical Trial Supply and Logistics Market: Revenue ($bn) by Region, 2017, 2022 and 2028
AbbVie Contract Pharmaceutical Manufacturing
Accucaps Industries Limited
ACM Global Central Lab
Agere Pharmaceuticals, Inc.
Allergan
Almac
AmerisourceBergen
Amgen
AndersonBrecon
Aptuit
Astellas Pharma
AstraZeneca
Bellwyck Packaging Solutions
BioClinica
Biotec Services International (Biotec)
Blackstone Group
Boehringer Ingelheim BioXcellence
Bristol-Myers Squibb
British Telecom (BT)
CAEN RFID
Cardinal Systems
Catalent Pharma Solutions
Celesio
Cenduit
Clínicos (CAIBER)
ClinIntel
ClinStar
COREX
Cortec
Cryoport
CSM
Custom Pharmaceuticals
DHL
Eli Lilly
EMD SERONO
EPS
ESMS
e-Trial Co.
Eurodifarm
Federal Express (FedEx)
Fisher Clinical Services
Frazier Healthcare
GE Healthcare
Greenbox
GSK
IBM
ICON
IMP Logistics
Intelleflex
Intelsius
Johnson & Johnson
Komtur Pharmaceuticals
LabCorp
LCAG USA
LifeConEx
Lonza
Marken
Mawdsleys
Medgenics
Merck & Co.
Micron Technologies
Millmount Healthcare
Mitsubishi Logistics
Mitsui & Co.
Movianto
Multipharma
NextPharma
Novo Nordisk
Novotech
N-SIDE
Nuvilex
Octapharma
Owens & Minor
Packaging Coordinators Inc. (PCI)
Parexel
Patheon N.V.
PCI Pharma Services
Perceptive Informatics
Pfizer
Pharmatek
Pieffe Group
PRA International
Quintiles
Recipharm
Redwood BioScience
Roche
Sanofi
SF Express
ShangPharma
Thermo Fisher Scientific
TNT Express
TransCelerate BioPharma
Tufts Center
UCB
United Parcel Service (UPS)
Vsoft Infoware
World Courier
Worldwide Clinical Trials
Y Prime
List of Organizations Mentioned in the Report
Agência Nacional de Vigilância Sanitária (ANVISA)
Central Drugs Standard Control Organization (CDSCO)
China FDA (CFDA)
Chinese Ministry of Health
Comissão Nacional de Ética em Pesquisa (CONEP)
Consorcio de Apoyo a la Investigación Biomédica – Plataforma Española de Ensayos Clínicos (CAIBER)
Drug Controller General of India (DCGI)
European Commission
European Medicines Agency (EMA)
Food and Drug Administration (FDA)
Health Science Authority
International Air Transport Association (IATA)
International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH)
International Pharmaceutical Federation
Japanese Ministry of Health, Labour and Welfare (MHLW)
Les Enterprises du Medicament (LEEM)
Medicines and Healthcare Products Regulatory Agency (MHRA)
National Institutes of Health (NIH)
Pharmaceuticals for Human Use (ICH)
Transportation Administration Security (TAS)
US Pharmacopeia (USP)
World Health Organization (WHO)
Download sample pages
Complete the form below to download your free sample pages for Clinical Trial Supply and Logistics Market for Pharma 2018-2028
Related reports
-

Biobanking Market Forecasts 2018-2028
Our 266-page report provides 145 tables, charts, clearly illustrating the data presented in this research. Read on to discover the...
Full DetailsPublished: 24 May 2018 -

Pharma Wholesale and Distribution Market Forecasts 2018-2028
The pharma wholesale and distribution market is estimated to grow at a CAGR of 5.6% in the first half of...Full DetailsPublished: 06 November 2018 -

Global Laboratory Information Systems Market Forecast 2018-2028
The global laboratory information systems market was is valued at $1.8bn in 2017 and is expected to grow at a...
Full DetailsPublished: 19 November 2018 -

Drug Discovery Outsourcing Market Forecast 2017-2027
Our 225-page report provides 150 tables, charts, and graphs. Read on to discover the most lucrative areas in the industry...
Full DetailsPublished: 13 October 2017 -

Global Lab Automation Market Forecast 2018-2028
The global lab automation market is expected to grow at a CAGR of 7.4% in the first half of the...Full DetailsPublished: 26 February 2018
Download sample pages
Complete the form below to download your free sample pages for Clinical Trial Supply and Logistics Market for Pharma 2018-2028
Do you have any custom requirements we can help you with?
Any specific country, geo region, market segment or specific company information?
Email us today, we can discuss your needs and see how we can help: jamie.roberts@visiongain.com
Would you like a free report overview of the report of your choice?
If so, please drop an email to Jamie Roberts stating your chosen report title to jamie.roberts@visiongain.com
Visiongain’s reports are based on comprehensive primary and secondary research. Those studies provide global market forecasts (sales by drug and class, with sub-markets and leading nations covered) and analyses of market drivers and restraints (including SWOT analysis) and current pipeline developments. To find out more about our reports methodology, please email jamie.roberts@visiongain.com
“Thank you for this Gene Therapy R&D Market report and for how easy the process was. Your colleague was very helpful and the report is just right for my purpose. This is the 2nd good report from Visiongain and a good price.”
Dr Luz Chapa Azuella, Mexico
American Association of Colleges of Pharmacy
American College of Clinical Pharmacy
American Pharmacists Association
American Society for Pharmacy Law
American Society of Consultant Pharmacists
American Society of Health-System Pharmacists
Association of Special Pharmaceutical Manufacturers
Australian College of Pharmacy
Biotechnology Industry Organization
Canadian Pharmacists Association
Canadian Society of Hospital Pharmacists
Chinese Pharmaceutical Association
College of Psychiatric and Neurologic Pharmacists
Danish Association of Pharmaconomists
European Association of Employed Community Pharmacists in Europe
European Medicines Agency
Federal Drugs Agency
General Medical Council
Head of Medicines Agency
International Federation of Pharmaceutical Manufacturers & Associations
International Pharmaceutical Federation
International Pharmaceutical Students’ Federation
Medicines and Healthcare Products Regulatory Agency
National Pharmacy Association
Norwegian Pharmacy Association
Ontario Pharmacists Association
Pakistan Pharmacists Association
Pharmaceutical Association of Mauritius
Pharmaceutical Group of the European Union
Pharmaceutical Society of Australia
Pharmaceutical Society of Ireland
Pharmaceutical Society Of New Zealand
Pharmaceutical Society of Northern Ireland
Professional Compounding Centers of America
Royal Pharmaceutical Society
The American Association of Pharmaceutical Scientists
The BioIndustry Association
The Controlled Release Society
The European Federation of Pharmaceutical Industries and Associations
The European Personalised Medicine Association
The Institute of Clinical Research
The International Society for Pharmaceutical Engineering
The Pharmaceutical Association of Israel
The Pharmaceutical Research and Manufacturers of America
The Pharmacy Guild of Australia
The Society of Hospital Pharmacists of Australia
Don’t Miss Out!
Subscribe to receive the latest Industry news, trending insight and analysis straight to your inbox.Choose your preferences:
Latest Pharma news
Retinal Gene Therapy Market
The global Retinal Gene Therapy market is projected to grow at a CAGR of 9.6% by 2034
26 July 2024
HIV Drugs and Injectables Market
The global HIV Drugs & Injectables market is projected to grow at a CAGR of 4.6 % by 2034
24 July 2024
Digital Twin Technology in Pharmaceutical Manufacturing Market
The global Digital Twin Technology in Pharmaceutical Manufacturing market is projected to grow at a CAGR of 31.3% by 2034
23 July 2024
Specialty Pharma Market
The global Specialty Pharma market is projected to grow at a CAGR of 7.5% by 2034
22 July 2024

















